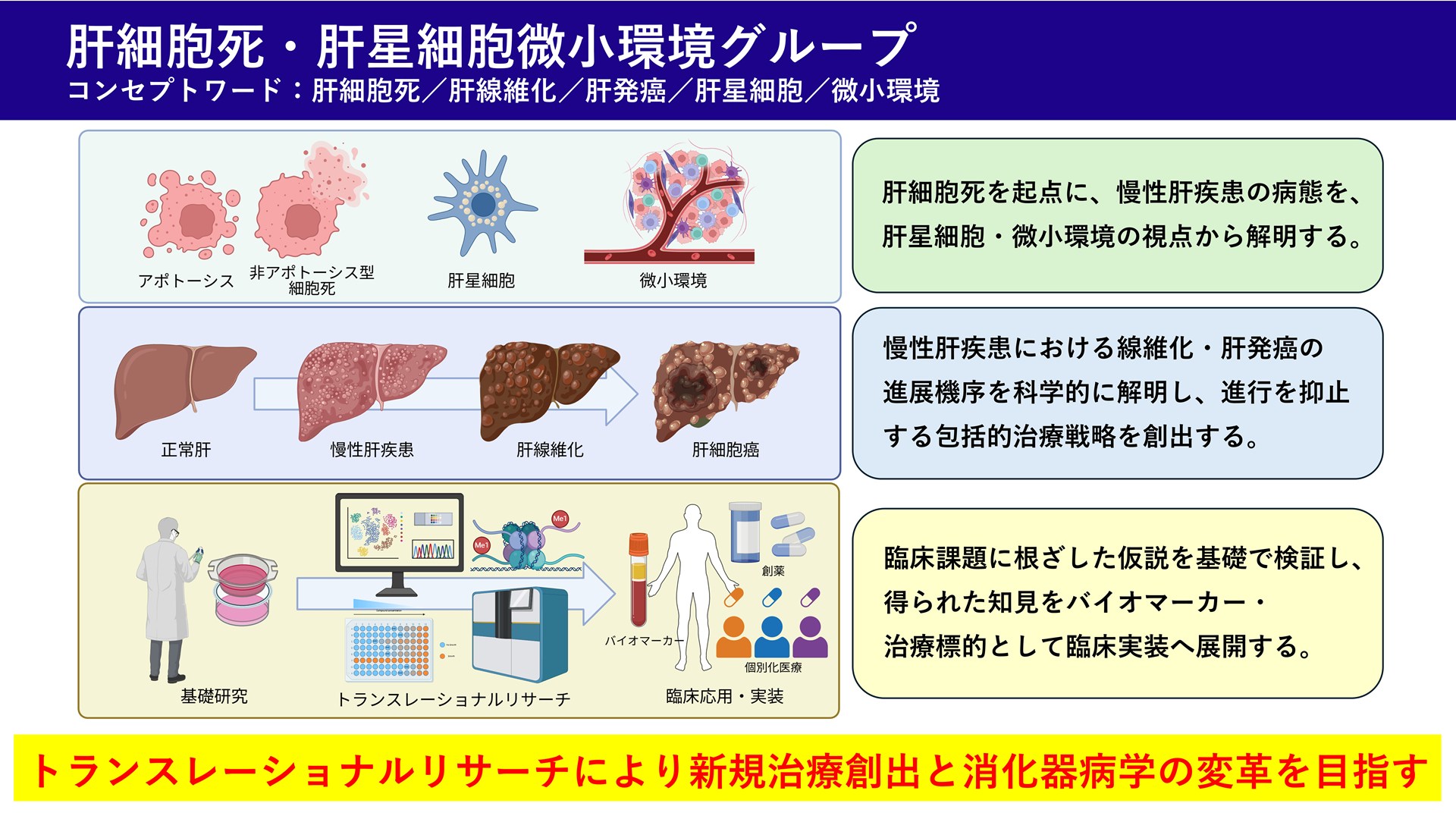
Hepatocyte Death and Hepatic Astrocyte Microenvironment Research Group
Group Leader: Yoshinobu Saito
Concept: To elucidate the pathological chain of chronic liver diseases from hepatocyte death to fibrosis and carcinogenesis through the interaction with the microenvironment centered on hepatic stellate cells, and to create novel therapies through translational research (bridge research).
The group is integrating the pathological chain of chronic liver diseases, starting from hepatocyte death (apoptotic and non-apoptotic types), inflammatory and immune responses, microenvironmental changes centered on hepatic astrocytes, fibrosis, and hepatocarcinogenesis. We combine single-cell RNA-seq, spatial transcriptomics, and chromatin analysis with mouse models, organoids, and co-culture systems to elucidate the function of hepatic astrocyte subpopulations, intercellular signals, and transcriptional regulatory networks. We will further validate these findings in clinical samples (biopsies, excised tissues, and blood), and through biomarker development and validation of therapeutic interventions, we aim to create novel preventive and therapeutic agents through translational research.
Elucidation of the pathological mechanism of chronic liver diseases initiated by hepatocyte death
We analyze how the type of hepatocyte death (apoptotic/non-apoptotic cell death) defines the pathogenesis of chronic liver disease and contributes to the development of fibrosis and carcinogenesis. We will systematically examine the spatiotemporal dynamics of receptor signals, cytokine/chemokine circuits, and immune cells (macrophages, neutrophils, T cells, etc.) activated by the release of DAMPs during cell death in our in vivo/in vitro model of hepatocyte death induction. We will elucidate the causal mechanism of disease progression. Furthermore, the obtained findings will be validated in clinical samples (biopsies, resected tissues, and blood) to identify biomarkers for predicting the progression of the disease and to create candidate therapeutic targets.
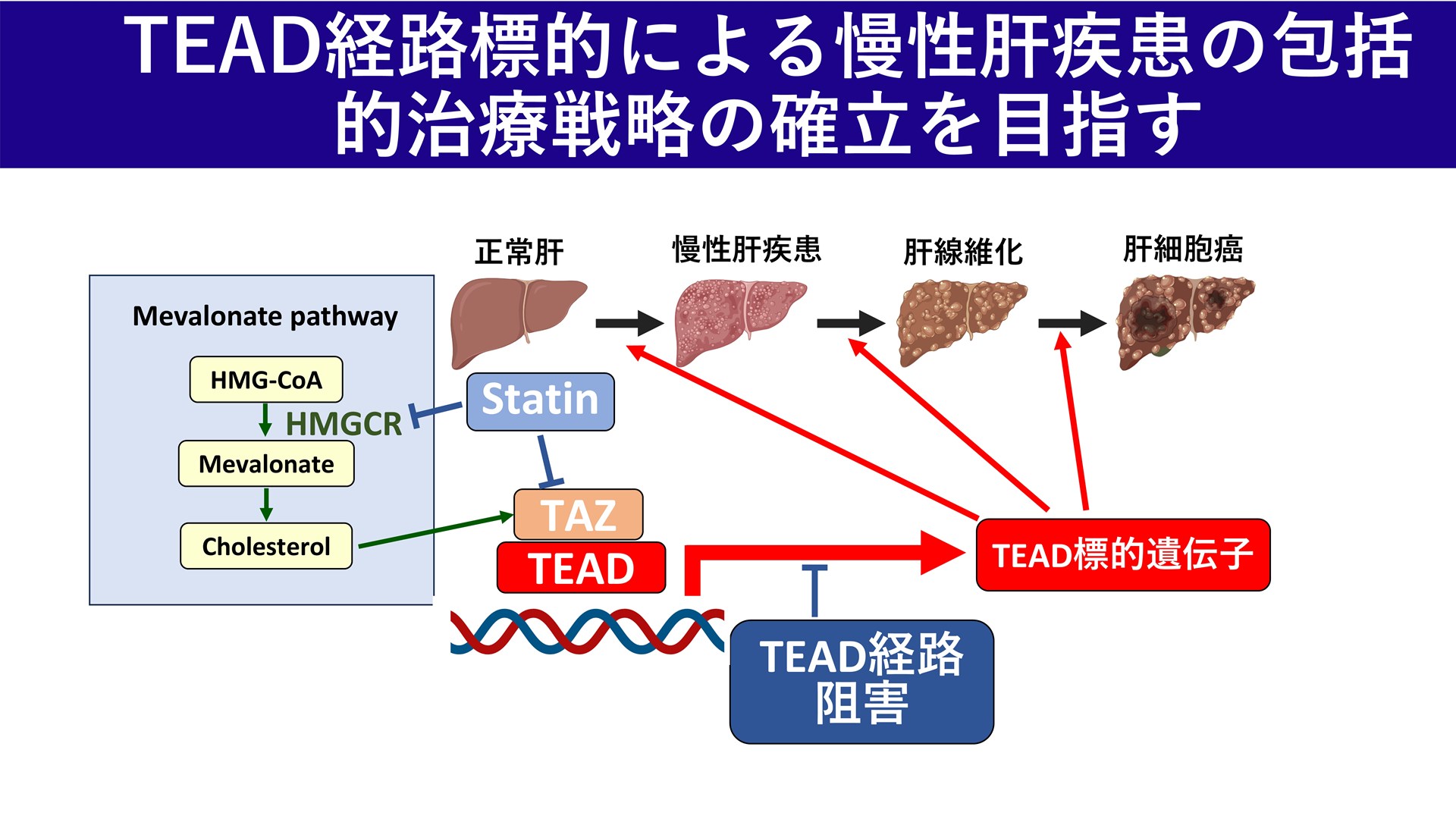
Establishment of a comprehensive treatment strategy for the development of liver fibrosis and hepatocellular carcinoma in chronic liver disease
Chronic liver disease is a continuum of pathological conditions that progresses from hepatocellular carcinoma to fibrosis via inflammatory and immune responses and hepatic astrocyte activation following hepatocyte death and other events. On the other hand, current therapies focus on interventions that target either fibrosis or hepatocellular carcinoma individually, and therapeutic systems that integrate and control the continuum of pathological conditions have not been fully established. Therefore, this study aims to establish a comprehensive treatment strategy to control inflammation and fibrosis in the background liver and to reduce the risk of carcinogenesis and recurrence within the same framework. Through identification of pathological drivers and validation of interventions, we aim to establish a novel treatment concept for prevention and early intervention.
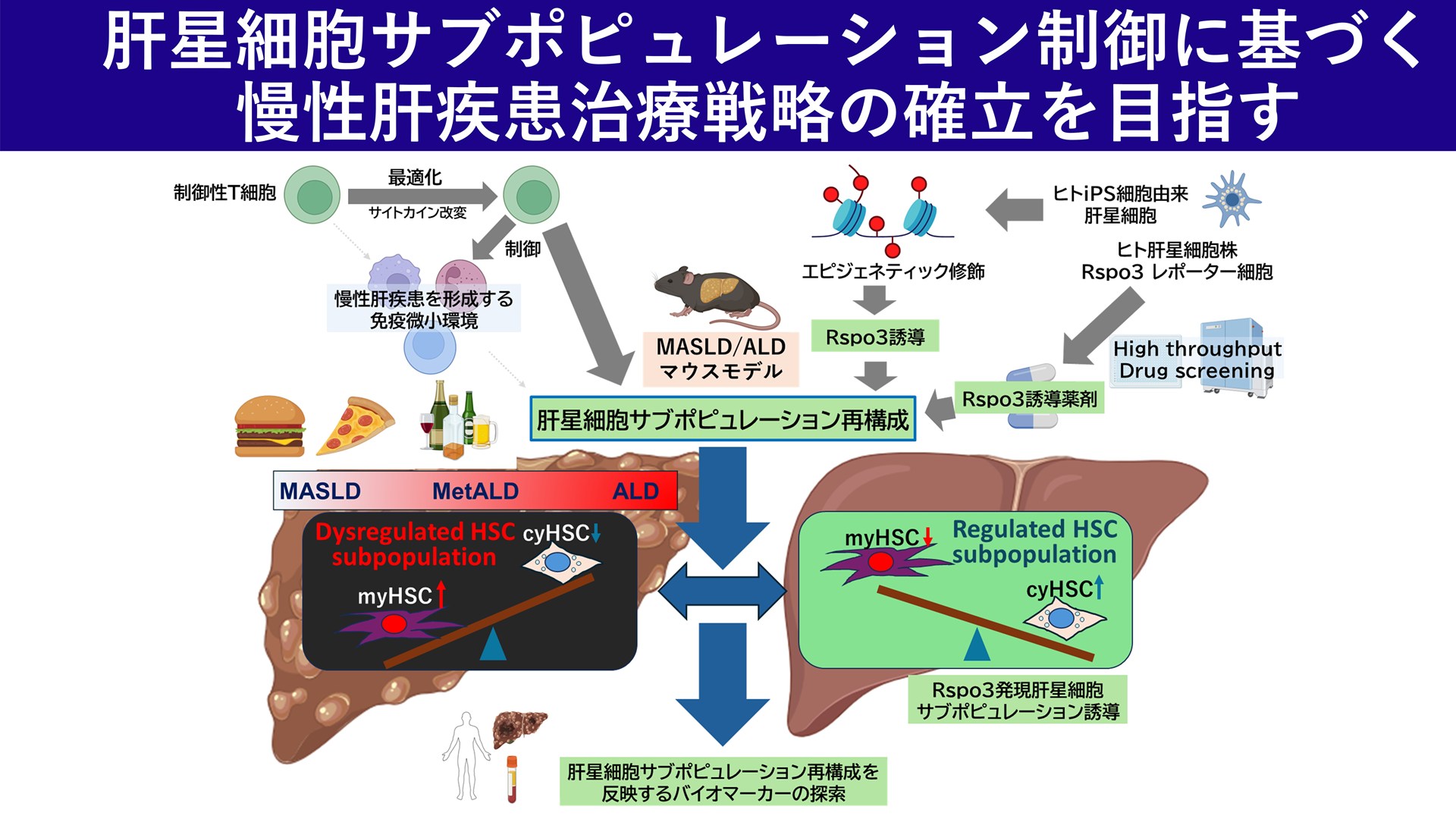
Establishment of therapeutic strategies for chronic liver disease by microenvironmental control focusing on hepatic stellate cell subpopulations.
Hepatic astrocytes are known to promote fibrosis and hepatocarcinogenesis through activation. Recently, we have shown that hepatic astrocytes consist of diverse subpopulations and that imbalance in their composition is associated with pathological progression and prognosis. In this study, we focus on the plasticity and population dynamics of hepatic stellate cells to identify the subpopulations and cell-cell interactions that drive fibrosis and carcinogenesis. We will visualize state transitions and localization of hepatic astrocytes by single-cell RNA-seq/spatial transcriptomics and reveal their signaling networks with immune cells, cholangiocytes, and vascular endothelium. Candidate factors will be analyzed for chromatin accessibility and enhancers, and their functions will be verified in co-culture and organoids. In addition, their contributions to extracellular substrate production, immunomodulation, and tumor niche formation will be quantified to identify targetable mechanisms and establish therapeutic strategies.
Future Prospects: Implementation of Personalized Medicine Based on Visualization and Stratification of Pathological Chains
In the future, we will quantify the "pathological chain" from both temporal (when it occurs) and spatial (where it occurs) aspects of the inflammatory and immune responses that start with hepatocyte death, restructure the microenvironment centered on hepatic stellate cells (HSCs), progress to fibrosis, and then to hepatocarcinogenesis, and identify the branching point where the disease stage moves to the next stage. A branching point is, for example, a point at which a switch in transcriptional regulatory networks or a rewiring of intercellular signals (cytokines, growth factors, extracellular matrix, immune checkpoints, etc.) occurs, and identifying this point is the key to prevention and early intervention. To this end, we integrate multi-layered omics analyses (single-cell/spatial analysis, chromatin analysis, proteomics, etc.) using clinical specimens (biopsy, excised tissue, blood) to construct a pathological map (multi-layered omics atlas). The information obtained will not be limited to research use, but will be incorporated into a test system that can be measured in the field (blood biomarkers, tissue scores, imaging and digital pathology indices, etc.) as indicators that ensure reproducibility and versatility. In addition, anti-fibrotic and anti-carcinogenic preventive/early intervention therapies will be developed by targeting the identified pathological drivers and branches. We will stratify patients by biomarker and develop clinical research that can verify "which intervention works for whom and when," and promote social implementation of diagnostic and therapeutic methods through industry-academia collaboration. Ultimately, we aim to establish a new medical model to deter the progression and recurrence of chronic liver diseases and contribute to the transformation of diagnosis and treatment.
Major Papers
- Kudo S, Hikita H, Saito Y, Murai K, Kodama T, Tatsumi T, Takehara T. Collaborative orchestration of BH3-only proteins governs Bak/Bax-dependent Cell Death Differ. 2025 Jun;32(6):1153-1166.
- Sugimoto A, Saito Y, Wang G, Sun Q, Yin C, Lee KH, Geng Y, Rajbhandari P, Hernandez C, Steffani M, Qie J, Savage T, Goyal DM, Ray KC, Neelakantan TV, Yin D, Melms J, Lehrich BM, Yasaka TM, Liu S, Oertel M, Lan T, Guillot A, Peiseler M, Filliol A, Kanzaki H, Fujiwara N, Ravi S, Izar B, Brosch M, Hampe J, Remotti H, Argemi J, Sun Z, Kendall TJ Sun Z, Kendall TJ, Hoshida Y, Tacke F, Fallowfield JA, Blockley-Powell SK, Haeusler RA, Steinman JB, Pajvani UB, Monga SP, Bataller R, Masoodi M, Arpaia N, Lee YA, Stockwell BR, Lee YA Lee YA, Stockwell BR, Augustin HG, Schwabe RF. Hepatic stellate cells control liver zonation, size and functions via R-spondin 3. Nature. 2025 Apr;640( 8059):752-761.
- Saito Y, Yin D, Kubota N, Wang X, Filliol A, Remotti H, Nair A, Fazlollahi L, Hoshida Y, Tabas I, Wangensteen KJ, Schwabe RF. TAZ-TEAD2 Pathway Drives the Growth of Hepatocellular Carcinoma via ANLN and KIF23. Gastroenterology. 2023 Jun;164(7):1279-1292.
- Filliol A, Saito Y, Nair A, Dapito DH, Yu LX, Ravichandra A, Bhattacharjee S, Affo S, Fujiwara N, Su H, Sun Q, Savage TM, Wilson-Kanamori JR, Caviglia JM, Chin L, Chen D, Wang X, Caruso S, Kang JK, Amin AD, Wallace S, Dobie R, Yin D, Rodriguez-Fiallos OM, Yin C Chin L, Chen D, Wang X, Caruso S, Kang JK, Amin AD, Wallace S, Dobie R, Yin D, Rodriguez-Fiallos OM, Yin C, Mehal A, Izar B, Friedman RA, Wells RG, Pajvani UB, Hoshida Y, Remotti HE, Arpaia N, Zucman-Rossi J, Karin M, Henderson NC, Tabas I, Schwabe RF. Opposing roles of hepatic stellate cell subpopulations in Nature. 2022 Oct;610(7931):356-365.
- Wang X, Zeldin S, Shi H, Zhu C, Saito Y, Corey KE, Osganian SA, Remotti HE, Verna EC, Pajvani UB, Schwabe RF, Tabas I. TAZ-induced Cybb contributes to liver J Hepatol. 2022 Apr;76(4):910-920.
- Affo S, Nair A, Brundu F, Ravichandra A, Bhattacharjee S, Matsuda M, Chin L, Filliol A, Wen W, Song X, Decker A, Worley J, Caviglia JM, Yu L, Yin D, Saito Y,. Savage T, Wells RG, Mack M, Zender L, Arpaia N, Remotti HE, Rabadan R, Sims P, Leblond AL, Weber A, Riener MO, Stockwell BR, Gaublomme J, Llovet JM, Kalluri R Michalopoulos GK, Seki E, Sia D, Chen X, Califano A, Schwabe RF. Promotion of cholangiocarcinoma growth by diverse cancer-associated fibroblast Cancer Cell. 2021 Jun 14;39(6):866-882. e11.
- Mizutani N, Hikita H, Saito Y, Myojin Y, Sato K, Urabe M, Kurahashi T, Shiode Y, Sakane S, Murai K, Nozaki Y, Kodama T, Sakamori R, Yoshida Y, Tatsumi T, Takehara T Takehara T. Gab1 in livers with persistent hepatocyte apoptosis has an anti-apoptotic effect and reduces chronic liver injury, fibrosis, and Am J Physiol Gastrointest Liver Physiol. 2021 Jun 1;320(6):G958-G968.
- Nozaki Y, Hikita H, Tanaka S, Fukumoto K, Urabe M, Sato K, Myojin Y, Doi A, Murai K, Sakane S, Saito Y, Kodama T, Sakamori R, Tatsumi T, Takehara T. Persistent hepatocyte apoptosis promotes tumorigenesis from diethylnitrosamine-transformed hepatocytes through increased oxidative stress, independent of Sci Rep. 2021 Feb 9;11(1):3363.
- Saito Y, Hikita H, Nozaki Y, Kai Y, Makino Y, Nakabori T, Tanaka S, Yamada R, Shigekawa M, Kodama T, Sakamori R, Tatsumi T, Takehara T. DNase II activated by the mitochondrial apoptotic pathway regulates RIP1-dependent non-apoptotic hepatocyte death via the TLR9/IFN-β signaling pathway. the mitochondrial apoptotic pathway regulates RIP1-dependent non-apoptotic hepatocyte death via the TLR9/IFN-β signaling pathway. Cell Death. Differ. 2019 Mar;26(3):470-486.
- Tanaka S, Hikita H, Tatsumi T, Sakamori R, Nozaki Y, Sakane S, Shiode Y, Nakabori T, Saito Y, Hiramatsu N, Tabata K, Kawabata T, Hamasaki M, Eguchi H, Nagano H Yoshimori T, Takehara T. Rubicon inhibits autophagy and accelerates hepatocyte apoptosis and lipid accumulation in nonalcoholic fatty liver disease in mice. Hepatology. 2016 Dec;64(6):1994-2014.
- Hikita H, Kodama T, Tanaka S, Saito Y, Nozaki Y, Nakabori T, Shimizu S, Hayashi Y, Li W, Shigekawa M, Sakamori R, Miyagi T, Hiramatsu N, Tatsumi T, Takehara T. Activation of the Mitochondrial Apoptotic Pathway Produces Reactive Oxygen Species and Oxidative Damage in Hepatocytes That Contribute to Liver Tumorigenesis. Cancer Prev Res (Phila). 2015 Aug;8(8):693-701.
- Kodama T, Hikita H, Kawaguchi T, Saito Y, Tanaka S, Shigekawa M, Shimizu S, Li W, Miyagi T, Kanto T, Hiramatsu N, Tatsumi T, Takehara T. The Bcl-2 homology domain 3 (BH3)-only proteins Bim and bid are functionally active and restrained by anti-apoptotic Bcl-2 family proteins in healthy liver. 2013 Oct 18;288(42):30009-30018.
- Hikita H, Kodama T, Shimizu S, Li W, Shigekawa M, Tanaka S, Hosui A, Miyagi T, Tatsumi T, Kanto T, Hiramatsu N, Morii E, Hayashi N, Takehara T. Bak deficiency J Hepatol. 2012 Jul;57(1):92-100.
- Hikita H, Takehara T, Kodama T, Shimizu S, Shigekawa M, Hosui A, Miyagi T, Tatsumi T, Ishida H, Li W, Kanto T, Hiramatsu N, Shimizu S, Tsujimoto Y, Hayashi N. Delayed-onset caspase-dependent massive hepatocyte apoptosis upon Fas activation in Bak/Bax-deficient mice. Hepatology. 2011 Jul;54(1):240-51.
- Hikita H, Takehara T, Shimizu S, Kodama T, Shigekawa M, Iwase K, Hosui A, Miyagi T, Tatsumi T, Ishida H, Li W, Kanto T, Hiramatsu N, Hayashi N. The Bcl-xL inhibitor, ABT-737, efficiently induces apoptosis and suppresses growth of hepatoma cells in combination with sorafenib. 2010 Oct;52(4):1310-21.
- Shimizu S, Takehara T, Hikita H, Kodama T, Miyagi T, Hosui A, Tatsumi T, Ishida H, Noda T, Nagano H, Doki Y, Mori M, Hayashi N. The let-7 family of microRNAs J Hepatol. 2010 May;52(5):698-704.
- Hikita H, Takehara T, Kodama T, Shimizu S, Hosui A, Miyagi T, Tatsumi T, Ishida H, Ohkawa K, Li W, Kanto T, Hiramatsu N, Hennighausen L, Yin XM, Hayashi N. BH3 Hepatology. 2009 Dec;50(6):1972-80.
- Hikita H, Takehara T, Shimizu S, Kodama T, Li W, Miyagi T, Hosui A, Ishida H, Ohkawa K, Kanto T, Hiramatsu N, Yin XM, Hennighausen L, Tatsumi T, Hayashi N. Mcl- 1 and Bcl-xL cooperatively maintain integrity of hepatocytes in developing and adult murine liver. Hepatology. 2009 Oct;50(4):1217-26.
- Takehara T, Tatsumi T, Suzuki T, Rucker EB 3rd, Hennighausen L, Jinushi M, Miyagi T, Kanazawa Y, Hayashi N. Hepatocyte-specific disruption of Bcl-xL Gastroenterology. 2004 Oct;127(4):1189-97.
