SLD Whole Body Network Research Group
Group Leader: Sadatsugu Sakane
Concept: Systemic Diseases / Multi-organ correlation / Borderless
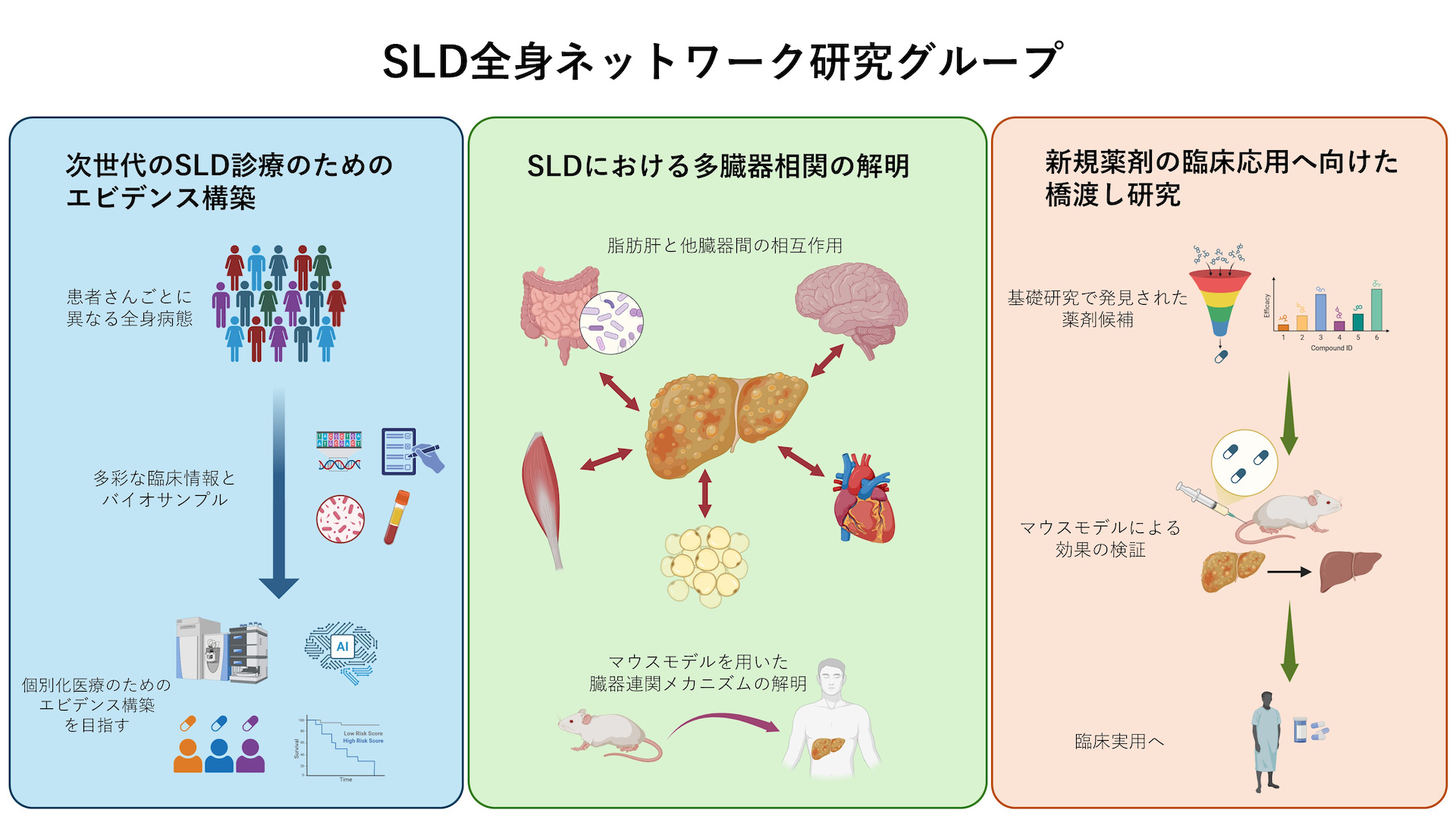
We consider SLD as one of the "systemic diseases" and create novel therapeutic strategies to improve the life prognosis of patients by elucidating the multi-organ linkage. We redefine fatty liver diseases (SLD) such as MASLD (metabolic dysfunction-related fatty liver disease) and ALD (alcohol-related liver disease) as "systemic diseases" with risk of cardiovascular diseases and cancer of other organs, and elucidate the network (multi-organ correlation) between the liver and all organs of the body beyond the boundaries of organs and methods. By integrating clinical data, results of basic experiments, and AI analysis, we aim to generate evidence that will serve as the foundation for the construction of a next-generation SLD treatment system that provides total coordination from prevention of disease onset to improvement of prognosis from the perspective of diagnosing not only the liver but also the entire body.
Building evidence for SLD precision medicine to achieve healthy longevity
SLD practice is now at a major turning point, led by the practical application of MASLD drugs overseas. However, since SLD is a systemic disease, it is essential to construct a new system of medical care that not only improves the local area of the liver but also achieves "healthy longevity" for patients in a holistic manner. We will accumulate sophisticated evidence through integrated analysis of multimodal data obtained from diverse clinical information and biosamples. By doing so, we aim to identify non-invasive diagnostic biomarkers, predict long-term prognosis, and create models for predicting the efficacy of therapeutic agents, which is the key to personalized medicine, and to implement precision medicine that can truly contribute to patients.
Elucidation of "organ correlation" by integrated analysis of human data and disease models
SLD (fatty liver disease) is a "systemic disease" that is closely associated with a wide variety of organs, including the intestine, brain, muscle, and adipose tissue. While liver disease increases the risk of cardiovascular disease and cancer of other organs, the condition of other organs also influences the development of liver disease. We seamlessly link findings from human clinical data to our own mouse models of disease to elucidate unknown molecular signals and intercellular networks that are exchanged between organs. By viewing the whole body as an organic system, not just the liver, we will identify "novel insights" into pathological conditions that could not be reached from conventional perspectives, and pioneer next-generation therapeutic strategies to improve the overall condition of the body.
Bridge research from identification of drug targets to clinical application
We will evaluate the efficacy and safety of molecular targets and therapeutic candidates discovered in basic research that contribute to the improvement of SLD using mouse models. In particular, we will examine the systemic benefits of treatment, taking into account spillover effects to other organs, and develop translational research that smoothly links the results of basic research to clinical research. Ultimately, through collaboration with pharmaceutical companies and academic institutions, we aim to provide new treatment options for patients suffering from SLD and its related complications.
Future Perspectives: Borderless Medicine: Next Generation SLD System Pioneered by Multi-Organ Network Analysis
The goal of this research group is to redefine SLD as a "systemic disease" rather than just a "liver disease," and to propose a new approach to medicine. We have adopted the concept of "Borderless" in order to remove the boundary between the liver and other organs, to unravel the whole body as a single dynamic network, and to eliminate the barriers between basic research and clinical practice so that knowledge can be circulated in both directions. We hope to link the biomarkers and therapeutic targets identified through the elucidation of this network to social implementation without delay, and to materialize a "next-generation SLD treatment system" that will enable precise prediction and intervention according to individual pathological conditions.
Major Papers
- Sakane S, Nishio T, Fuji H, Park SY, Ishizuka K, Miciano C, Kimura Y, Hosseini M, Diggle K, Zhang V, Lee W, Kim HY, Liu X, Wang A, Brenner DA, Kisseleva T. Msln/ Muc16 signaling in activated portal fibroblasts drives the development of cholestatic fibrosis and HCC in aged female Mdr2-/- mice. Gastroenterol Hepatol. 2026 Apr 10:101785. doi: 10.1016/j.jcmgh.2026.101785
- Kogiso T, Akuta N, Ikejima K, Otsuka M, Kawaguchi T, Kawanaka M, Takahashi H, Tamaki N, Nakagawa H, Hikita H, Fujii H, Yamaguchi K, Yoneda M, Kon K, Uchiyama A M, Seko Y, Sakane S, Shigefuku R, Fujiwara N, Iwaki M, Kobayashi T, Adachi T, Takeuchi Y, Tsutsumi T, Nakano, Shibayama K, Urata N, Miyaaki H, Kuroda H, Koseki M, Sone H, Matsubayashi Y, Kakisaka K, Takai A, Notsumata K, Seike M, Takei Y, Takeyama Y, Eguchi S, Watanabe S, Isomoto H, Yotsuyanagi H, Itoi T, Takehara T, Mochida S. Mochida S. Evidence-based clinical practice guidelines for metabolic dysfunction-associated steatotic liver disease (MASLD) 2026. Hepatol Res. in press
- Li D, Loneker AE, Safraou Y, Ford J, Mihelc E, Sakane S, Kisseleva T, Levental KR, Levental I, Sack I, Janmey PA, Wells RG. Cholesterol-containing lipid crystals can directly stiffen the rat steatotic liver before fibrosis. Proc Natl Acad Sci U S A. 2026 Jan 13;123(2):e2518060123.
- Ganguly S, Sakane S, Hokutan K, Zhang V, Miciano C, Wang A, Brenner DA, Kisseleva T. Aging and Aging-Related Senescence in the Liver. Semin Liver Dis. 2025 Dec;. 45(4):549-566.
- Yamamoto G, Carvalho-Gontijo Weber R, Lee W, Zhang V, Jang H, Sakane S, Liu X, Kim HY, Brenner DA, Li N, Kisseleva T. DHCR7 inhibition ameliorates MetALD and HCC in mice and human 3D liver spheroids. JHEP Rep. 2025 Apr 5;7(7):101415.
- Sakane S, Hikita H. Revisiting the relationship between thyroid function and metabolic dysfunction-associated steatotic liver disease in the era of J Gastroenterol. 2025 Mar;60(3):389-391.
- Gu L, Zhu Y, Nandi SP, Lee M, Watari K, Bareng B, Ohira M, Liu Y, Sakane S, Carlessi R, Sauceda C, Dhar D, Ganguly S, Hosseini M, Teneche MG, Adams PD, Gonzalez DJ Kisseleva T; Liver Cancer Collaborative; Tirnitz-Parker JEE, Simon MC, Alexandrov LB, Karin M. FBP1 controls liver cancer evolution from senescent Nature. 2025 Jan;637(8045):461-469.
- Sakane S, Hikita H, Shirai K, Sakamoto T, Narumi R, Adachi J, Kakita N, Yamada Y, Toyoda H, Takahashi H, Suda G, Kai M, Tahata Y, Sakamori R, Kumazaki S, Fukumoto K, Myojin Y, Murai K, Kodama T, Tatsumi T, Tomonaga T, Sakamoto N, Morii E, Takehara T. Fukumoto K, Myojin Y, Murai K, Kodama T, Tatsumi T, Tomonaga T, Sakamoto N, Morii E, Takehara T. Proteomic analysis of serum extracellular vesicles reveals Fibulin-3 as a Proteomic analysis of serum extracellular vesicles reveals Fibulin-3 as a new marker predicting liver-related events in MASLD. Hepatol Commun. 2024 Jun 3;8(6):e0448.
- Osna NA, Tikhanovich I, Ortega-Ribera M, Mueller S, Zheng C, Mueller J, Li S, Sakane S, Weber RCG, Kim HY, Lee W, Ganguly S, Kimura Y, Liu X, Dhar D, Diggle K Brenner DA, Kisseleva T, Attal N, McKillop IH, Chokshi S, Mahato R, Rasineni K, Szabo G, Kharbanda KK. Alcohol-Associated Liver Disease Outcomes: Critical Mechanisms of Critical Mechanisms of Liver Injury Progression. Biomolecules. 2024 Mar 27;14(4):404.
- Isaac R, Bandyopadhyay G, Rohm TV, Kang S, Wang J, Pokhrel N, Sakane S, Zapata R, Libster AM, Vinik Y, Berhan A, Kisseleva T, Borok Z, Zick Y, Telese F, Webster NJG, Olefsky JM. TM7SF3 controls TEAD1 splicing to prevent MASH-induced liver fibrosis. Cell Metab. 2024 May 7;36(5):1030-1043.e7.
- Kim HY, Sakane S, Eguileor A, Carvalho Gontijo Weber R, Lee W, Liu X, Lam K, Ishizuka K, Rosenthal SB, Diggle K, Brenner DA, Kisseleva T. The Origin and Fate of Cell Mol Gastroenterol Hepatol. 2024;17(1):93-106.
- Liu X, Lam K, Zhao H, Sakane S, Kim HY, Eguileor A, Diggle K, Wu S, Gontijo Weber RC, Soroosh P, Hosseini M, Mekeel K, Brenner DA, Kisseleva T. Isolation of primary human liver cells from normal and nonalcoholic steatohepatitis livers. STAR Protoc. 2023 Sep 15;4(3):102391.
- Sakane S, Hikita H, Shirai K, Myojin Y, Sasaki Y, Kudo S, Fukumoto K, Mizutani N, Tahata Y, Makino Y, Yamada R, Kodama T, Sakamori R, Tatsumi T, Takehara T. White Adipose Tissue Autophagy and Adipose-Liver Crosstalk Exacerbate Nonalcoholic Fatty Liver Disease in Mice. ;12(5):1683-1699.
