Liver Disease Clinical Research Group
Group Leader: Yuuki Tabata
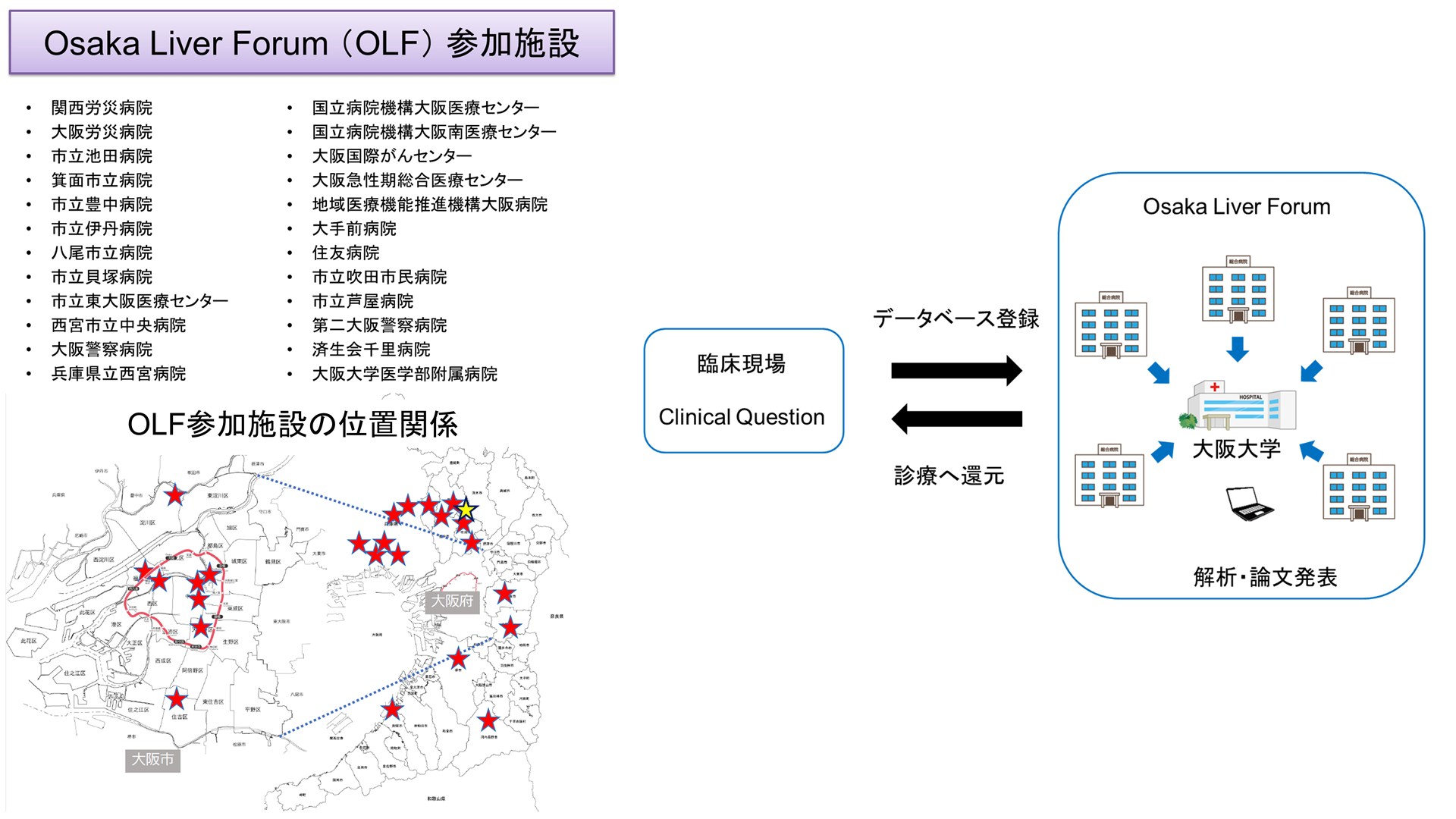
Concept: Real-World Evidence - Verify questions in daily medical practice using multicenter data and return the results to medical practice.
Our philosophy is to take clinical questions about liver diseases that arise in daily medical practice as a starting point, sublimate them into evidence through clinical research, and return the results to medical practice. Based on the Osaka Liver Forum (OLF), which consists of Osaka University and affiliated institutions, we will promote the evaluation and stratification of long-term outcomes, treatment efficacy, safety, and carcinogenic risk in hepatitis B and C, fatty liver disease (MASLD/MetALD), alcohol-related liver disease, liver cirrhosis, and hepatocellular carcinoma. The project will also include the following In addition to clinical information, stored serum and liver tissue of some patients will be used to elucidate the pathophysiology and search for new biomarkers in an integrated manner.
Long-term Outcomes and Liver Carcinogenesis Risk Stratification after Antiviral Treatment in Hepatitis B and C
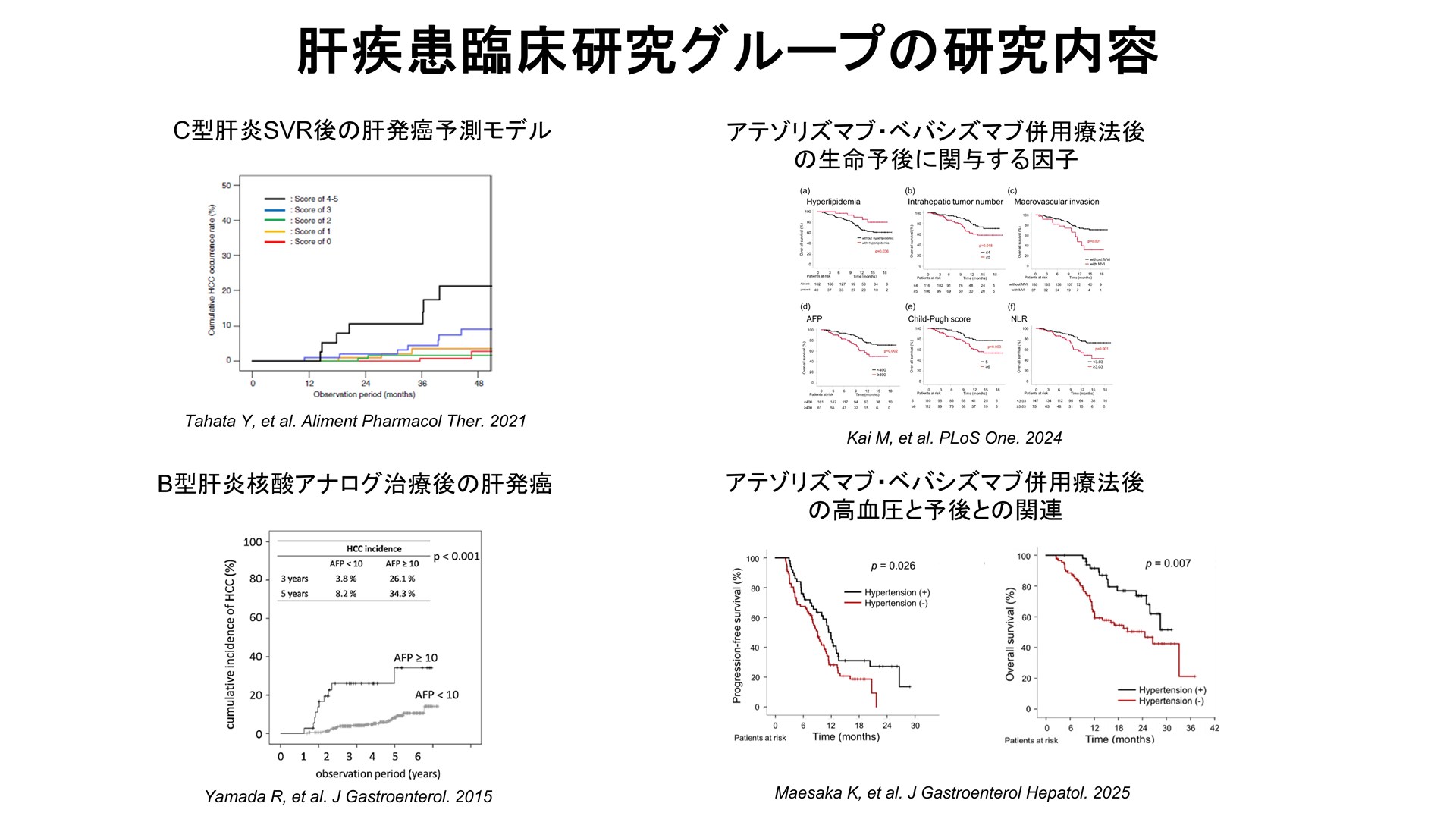
In OLF, the rate of first liver cancer in hepatitis C patients who achieved SVR with DAA therapy was 4.41 TP3T at 4 years, and the rate of first liver cancer in hepatitis C patients who achieved SVR with DAA therapy was 4.41 TP3T at 4 years. The rate was 4.41 TP3T at 4 years, with a BMI (≥ 25 kg/m2), FIB-4 index (≥ 3.25), albumin level at SVR (< 4.0 g/dl) and AFP level at SVR (≥ 5 ng/ml) were risk factors. We also found that a prediction score using those risk factors allowed stratification of carcinogenic risk1). In hepatitis B, the first liver cancer rate was 17.21 TP3T at 7 years in patients whose HBV-DNA was controlled by nucleic acid analog therapy, and age (≥ 55 years), cirrhosis, and AFP level (≥ 10 ng/ml) at 24 weeks of treatment were risk factors2 ). The findings obtained will be used to identify high-risk groups, lead to optimization of surveillance intervals and intervention points, and disseminated as an implementable stratification model. 1) Tahata Y, et al. Aliment Pharmacol Ther. 2021 2)Yamada R, et al. J Gastroenterol. 2015
Real-World Evidence of Drug Therapy Effectiveness for Hepatocellular Carcinoma and Prediction of Treatment Response
The drug therapy for hepatocellular carcinoma (HCC) is diversified, and it is important to optimize it based on treatment efficacy, adverse events, and liver function trends in real-life clinical practice. In patients with unresectable hepatocellular carcinoma treated with atezolizumab/bevacizumab, the following factors were reported to be associated with poor prognosis: no dyslipidemia, number of intrahepatic tumors (≥ 5), vascular invasion, AFP level (≥ 400ng/ml), Child-Pugh score (≥ 6) and NLR (≥ 3.03) 3). We also reported that patients with unresectable hepatocellular carcinoma who received atezolizumab-bevacizumab combination therapy and had adverse events of hypertension within 9 weeks of treatment had a favorable prognosis4) . We will further explore and validate biomarker candidates that can stratify reactivity/toxicity/prognosis using stored serum and liver tissue, and investigate the clinical applicability of indicators that can contribute to treatment selection. This will also lead to optimization of treatment sequences and follow-up plans. (3) Kai M, et al. PLoS One. 2024 4) Maesaka K, et al. J Gastroenterol Hepatol. 2025
Factors and risk stratification for the development of fatty liver disease (MASLD/MetALD) and alcohol-related liver disease
The number of patients with fatty liver disease and alcohol-related liver disease is increasing, and the pathways leading to fibrosis progression and carcinogenesis are diverse. We are evaluating factors associated with fibrosis progression, liver-related events, and carcinogenesis based on daily medical information such as metabolic abnormalities, drinking patterns, weight changes, and comorbidities, and are constructing a risk stratification model. At the same time, using stored serum and some liver tissues, we are searching for and validating candidate indicators to detect the progression of fibrosis at an early stage, aiming to develop an evaluation method that will contribute to the optimization of drug therapy, which is expected to be approved in the future. In addition, we will incorporate changes in test values and liver hardness over time to identify high-risk groups for progression of the disease and to propose follow-up intervals. We will also perform metabolomic analysis of stored serum to extract candidate markers that reflect the pathological condition.
Prediction of Noncompensated Events in Cirrhosis and Search for Indicators to Improve Prognosis
In cirrhosis, uncompensated events such as ascites, variceal hemorrhage, and hepatic encephalopathy have a significant prognostic impact, and OLF is conducting a prospective observational study of patients with cirrhosis to evaluate factors related to event occurrence, rehospitalization, and death by integrating the impact of liver reserve index, inflammation and coagulation, renal function, and complication treatment, Risk stratification and optimization of follow-up strategies for decision making. Furthermore, using stored serum and liver tissue, we will search for and validate new biomarker candidates that anticipate decompensation and worsening prognosis, leading to implementable risk assessment.
Future Prospects: From "Supporting Medical Practice" to "Changing Medical Practice" - Creating New Evidence through Domestic and International Collaboration and Intervention Research
To date, OLF has continuously disseminated Real-World Evidence to support routine medical care based on multicenter prospective observational studies. Going forward, OLF will further develop this solid foundation, strengthen ties with collaborators in Japan and overseas, and aim to generate highly universal findings through international collaborative research. In addition, we will take the hypotheses obtained through observational studies as a starting point and expand them into intervention studies to prospectively verify the optimization of medical treatment processes and strategies, thereby taking on the challenge of generating evidence that will actually change medical treatment. We will also integrate biomarker research utilizing preserved serum and liver tissue to accelerate the implementation of stratified medicine and its return to patients and society.
Major Papers
- Tahata Y, Hikita H, Takaki A, Kurosaki M, Matsuura K, Yatsuhashi H, Kuroda H, Ueno Y, Maekawa S, Nakamura M, Sakamori R, Kodama T, Takehara T. Long-term changes in hepatic reserve and prognosis after direct-acting antiviral treatment in patients with hepatitis C virus-related decompensated cirrhosis J Gastroenterol. 2025 Oct 30. doi: 10.1007/s00535-025-02312-1. Epub ahead of print. PMID: 41168423.
- Tatsumi N, Matsumoto K, Ishida H, Iio S, Nawa T, Kakita N, Nakahara M, Hosui A, Yoshida Y, Usui T, Imanaka K, Doi Y, Sakakibara M, Takehara T. Comparative analysis of lenvatinib use after atezolizumab plus bevacizumab versus lenvatinib as first-line therapy in unresectable hepatocellular carcinoma. Gastroenterol. 2026 Jan;61(1):68-77. doi: 10.1007/s00535-025-02308-x. Epub 2025 Oct 25. PMID: 41139139.
- Maesaka K, Hikita H, Kai M, Tahata Y, Shirai K, Doi A, Murai K, Saito Y, Yamada R, Kodama T, Ohkawa K, Miyazaki M, Ishida H, Matsumoto K, Nozaki Y, Yakushijin T, Sakamori R, Tatsumi N, Iio S, Nawa T, Kakita N, Nakahara M, Hosui A, Usui T, Imanaka K, Doi Y, Sakakibara Sakamori R, Tatsumi N, Iio S, Nawa T, Kakita N, Nakahara M, Hosui A, Usui T, Imanaka K, Doi Y, Sakakibara M, Yoshida Y, Tatsumi T, Takehara T. Hypertension as an Adverse Event Potentially Impacting Hypertension as an Adverse Event Potentially Impacting the Therapeutic Efficacy of Atezolizumab Plus Bevacizumab in Patients With Unresectable Hepatocellular J Gastroenterol Hepatol. 2025 Oct;40(10):2580-2590. doi: 10.1111/jgh.70068. Epub 2025 Sep 9. PMID: 40922653; PMCID: PMC12511841.
- Miyaaki H, Ueno Y, Itoh Y, Matsuura K, Takami T, Asahina Y, Suda G, Akuta N, Tateishi R, Nakamoto Y, Kakazu E, Terai S, Shimizu M, Miyazaki M, Nozaki Y, Sobue S, Yano H, Miyaki T, Moriuchi A, Hori T, Shirai K, Murai K, Saito Y, Kodama T, Tatsumi T, Yamada T, Takehara T. Factors involved in gastroesophageal varix Factors involved in gastroesophageal varix- related events in patients with hepatitis C virus-related compensated and decompensated cirrhosis after direct-acting antiviral therapy. Res. 2024 Oct 29. doi: 10.1111/hepr.14131. Epub ahead of print. PMID: 39470448.
- Kakita N, Morishita N, Hiramatsu N, Usui T, Imanaka K, Doi Y, Sakakibara M, Yoshida Y, Oze T, Kodama T, Tatsumi T, Takehara T. Clinical factors associated with the therapeutic efficacy of atezolamide Clinical factors associated with the therapeutic efficacy of atezolizumab plus bevacizumab in patients with unresectable hepatocellular carcinoma: A multicenter prospective PLoS One. 2024 Jan 2;19(1):e0294590. doi: 10.1371/journal.pone.0294590. PMID: 38165900; PMCID: PMC10760712.
- Tahata Y, Hikita H, Mochida S, Enomoto N, Ido A, Kuroda H, Miki D, Kurosaki M, Hiasa Y, Sakamori R, Kawada N, Yamashita T, Suda G, Yatsuhashi H, Yoshiji H, Kato N, Takami T, Nakao K, Matsuura K, Asahina Y, Itoh Y, Tateishi R, Nakamoto Y, Kakazu E, Terai S, Shimizu M, Ueno Y, Akuta N, Miyazaki M, Nozaki Y, Kabayama M, Moriuchi A Sobue S, Moriuchi A, Miyaki T, Kodama T, Tatsumi T, Yamada T, Takehara T. Posttreatment liver function, but not baseline liver function stratifies Moriuchi A, Miyaki T, Kodama T, Tatsumi T, Yamada T, Takehara T. Posttreatment liver function, but not baseline liver function stratifies patient survival after direct-acting antiviral treatment in decompensated cirrhosis with hepatitis C virus. Epub 2023 Oct 13. PMID: 37831182; PMCID: PMC10657790.
- Maesaka K, Sakamori R, Yamada R, Doi A, Tahata Y, Ohkawa K, Oshita M, Miyazaki M, Yakushijin T, Nozaki Y, Matsumoto K, Tanaka S, Kaneko A, Iio S, Nawa T, Yamada Y, Morishita N, Usui T, Hiramatsu N, Doi Y, Sakakibara M, Imanaka K, Yoshida Y, Kodama T, Hikita H, Tatsumi T, Takehara T. Pretreatment with antibiotics is associated with reduced therapeutic response to atezidime PLoS One. 2023 Feb 7;18(2): e0281459. doi e0281459. doi: 10.1371/journal.pone.0281459. PMID: 36749777; PMCID: PMC9904470.
- Tahata Y, Sakamori R, Yamada R, Kodama T, Hikita H, Hagiwara H, Oshita M, Imai Y, Hiramatsu N, Mita E, Kaneko A, Miyazaki M, Ohkawa K, Hijioka T, Fukui H, Ito T, Yamamoto K, Doi Y, Yoshida Y, Yamada Y, Yakushijin T, Tatsumi T, Takehara T; Osaka Liver Forum. Yamamoto K, Doi Y, Yoshida Y, Yamada Y, Yakushijin T, Tatsumi T, Takehara T; Osaka Liver Forum. Response Enhanced Prognosis in Hepatitis C with Compensated Advanced Liver Fibrosis. Dig Dis Sci. 2023 May;68(5):2115-2122. doi: 10.1007/s10620-022 -07629-y. Epub 2022 Dec 16. PMID: 36526814.
- Ohkawa K, Yakushijin T, Sakakibara M, Iio S, Doi Y, Tatsumi T, Takehara T. Effect of sofosbuvir and velpatasvir therapy on clinical outcome in hepatitis C Hepatol Res. 2023 Apr;53(4):301-311. doi: 10.1111/hepr.13868. Epub 2022 Dec 22. PMID: 36507871.
- Tahata Y, Sakamori R, Yamada R, Kodama T, Hikita H, Nozaki Y, Oshita M, Hiramatsu N, Miyazaki M, Mita E, Yamamoto K, Ohkawa K, Kaneko A, Ito T, Doi Y, Takehara T. Yakushijin T, Hijioka T, Fukui H, Imanaka K, Yoshida Y, Yamada Y, Tatsumi T, Takehara T. Risk of hepatocellular carcinoma after sustained virologic Hepatol Res. 2022 Oct;52(10):824-832. doi: 10.1111/hepr.13806. Epub 2022 Jul 8. Epub 2022 Jul 8. PMID: 35749289.
- Shinkai K, Sakamori R, Yamada R, Tahata Y, Nozaki Y, Matsumoto K, Tawara S, Fukuda K, Yoshida Y, Tanaka S, Ito T, Doi Y, Iio S, Sakakibara M, Nakanishi F, Takehara T. Kodama T, Hikita H, Tatsumi T, Takehara T. Prognostic impact of worsening of esophageal varices after balloon-occluded retrograde transvenous J Gastroenterol Hepatol. 2022 Jun;37(6):1148-1155. doi: 10.1111/jgh.15853. Epub 2022 Apr 26. PMID: 35430734.
- Maesaka K, Sakamori R, Yamada R, Doi A, Tahata Y, Miyazaki M, Ohkawa K, Mita E, Iio S, Nozaki Y, Yakushijin T, Imai Y, Kodama T, Hikita H, Tatsumi T, Takehara T. Comparison of atezolizumab plus bevacizumab and lenvatinib in terms of efficacy and safety as primary systemic chemotherapy for hepatocellular Hepatol Res. 2022 Jul;52(7):630-640. doi: 10.1111/hepr.13771. Epub 2022 Apr 25. PMID: 35417606.
- Sakamori R, Yamada R, Tahata Y, Kodama T, Hikita H, Tatsumi T, Yamada T, Takehara T. The absence of warfarin treatment and situs inversus are associated with the occurrence of hepatocellular carcinoma after Fontan surgery. J Gastroenterol. 2022 Feb;57(2):111-119. doi: 10.1007/s00535-021-01842-8. Epub 2022 Jan 22. PMID: 35064829.
- Yamada R, Kodama T, Tatsumi T, Yamada T, Takehara T. Liver-related events after direct-acting antiviral therapy in patients with hepatitis C virus- associated cirrhosis. J Gastroenterol. 2022 Feb;57(2):120-132. doi: 10.1007/s00535-021-01845-5. Epub 2022 Jan 20. PMID: 35059853.
- Maesaka K, Sakamori R, Yamada R, Tahata Y, Imai Y, Ohkawa K, Miyazaki M, Mita E, Ito T, Hagiwara H, Yakushijin T, Kodama T, Hikita H, Tatsumi T, Takehara T. Hyperprogressive disease in patients with unresectable hepatocellular carcinoma receiving atezolizumab plus bevacizumab therapy. 2022 Mar;52(3):298-307. doi: 10.1111/hepr.13741. Epub 2021 Dec 28. Erratum in: Hepatol Res. 2022 Jul;52(7):659. doi: 10.1111/hepr.13775. PMID: 34918428. 34918428.
- Tahata Y, Sakamori R, Yamada R, Kodama T, Hikita H, Hagiwara H, Imai Y, Hiramatsu N, Tamura S, Yamamoto K, Oshita M, Ohkawa K, Hijioka T, Fukui H, Ito T, Doi Y, Yamada Y, Yakushijin T, Yoshida Y, Tatsumi T, Takehara T; Osaka Liver Forum. Prediction model for hepatocellular carcinoma occurrence in patients with Aliment Pharmacol Ther. 2021 Nov;54(10):1340-1349. doi: 10.1111/apt.16632. Epub 2021 Oct 7. PMID : 34618934. Epub 2021 Oct 7. doi: 10.1111/apt.16632.
- Maesaka K, Sakamori R, Yamada R, Tahata Y, Ohkawa K, Oshita M, Tamura S, Hagiwara H, Mita E, Yakushijin T, Inada M, Kodama T, Hikita H, Tatsumi T, Takehara T. Efficacy of Ramucirumab Versus Sorafenib as Subsequent Treatment for Hepatocellular Carcinoma. Anticancer Res. 2021 Apr;41(4):2187-2192. doi: 10. PMID: 33813432.
- Maesaka K, Sakamori R, Yamada R, Tahata Y, Ohkawa K, Oshita M, Tamura S, Hagiwara H, Mita E, Yakushijin T, Inada M, Kodama T, Hikita H, Tatsumi T, Takehara T. Efficacy of Ramucirumab Versus Sorafenib as Subsequent Treatment for Hepatocellular Carcinoma. Anticancer Res. 2021 Apr;41(4):2187-2192. doi: 10. PMID: 33813432.
- Maesaka K, Sakamori R, Yamada R, Tahata Y, Ohkawa K, Oshita M, Tamura S, Hagiwara H, Mita E, Yakushijin T, Inada M, Kodama T, Hikita H, Tatsumi T, Takehara T. Efficacy of Ramucirumab Versus Sorafenib as Subsequent Treatment for Hepatocellular Carcinoma. Anticancer Res. 2021 Apr;41(4):2187-2192. doi: 10. PMID: 33813432.
- Itoh Y, Yamashita T, Kanto T, Suda G, Nakamoto Y, Kato N, Asahina Y, Matsuura K, Terai S, Nakao K, Shimizu M, Takami T, Akuta N, Yamada R, Kodama T, Tatsumi T, Yamada T, Takehara T Yamada T, Takehara T. Sofosbuvir plus velpatasvir treatment for hepatitis C virus in patients with decompensated cirrhosis: a Japanese real-world J Gastroenterol. 2021 Jan;56(1):67-77. doi: 10.1007/s00535-020-01733-4. Epub 2020 Oct 1. PMID: 33001338.
