Hepatitis B Virus (HBV) Research Group
Group Leader: Kazuhiro Murai
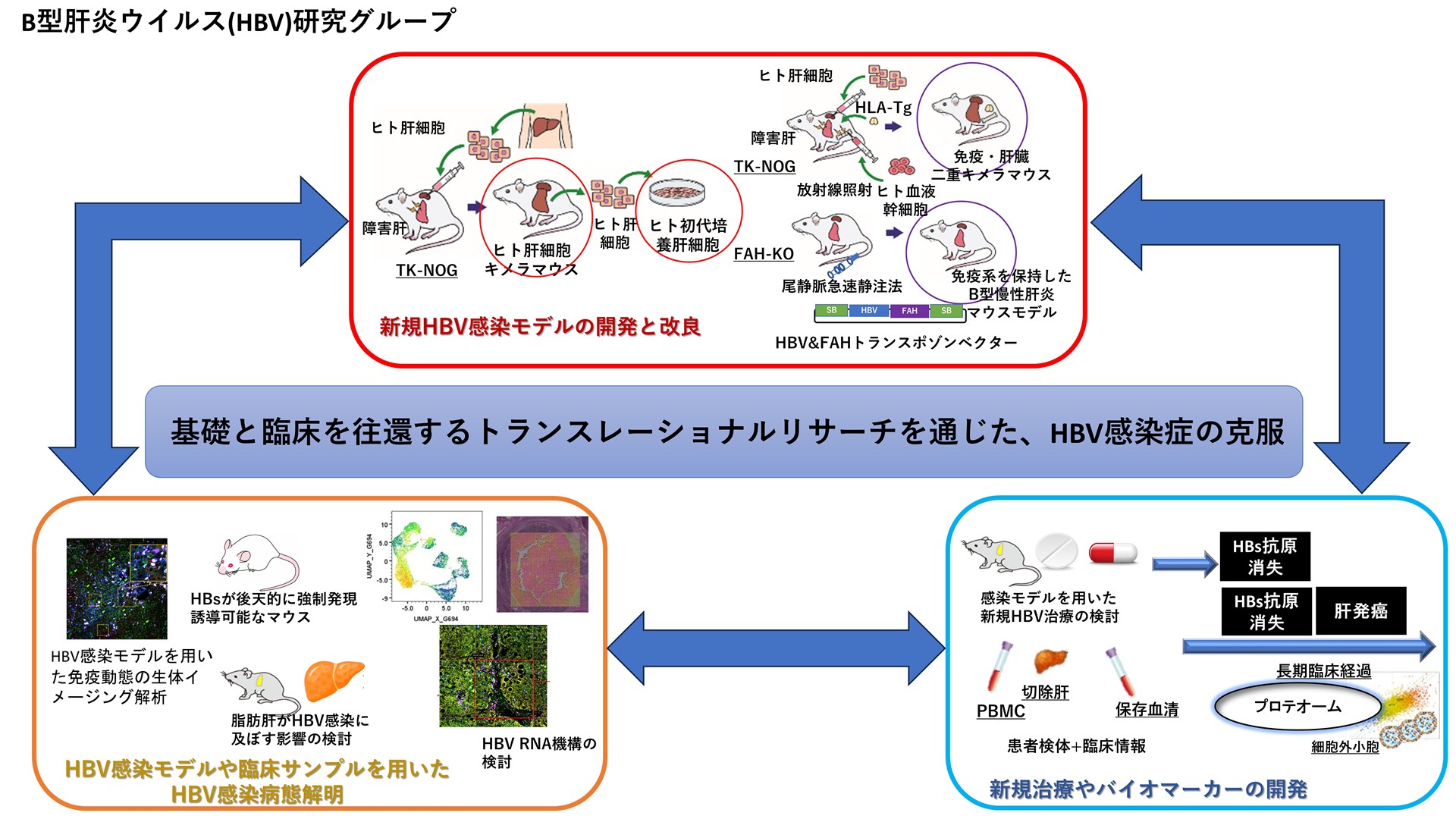
Concept: Aiming for complete elimination of HBV
Hepatitis B virus (HBV) infects approximately 250 million people worldwide, and chronic persistent infection can progress to hepatitis, cirrhosis, and hepatocellular carcinoma (HCC). Although nucleic acid analogs (NUCs) have enabled strong suppression of HBV replication, the presence of covalently closed circular DNA (cccDNA) formed in the nuclei of infected hepatocytes and the HBV genome integrated into the host genome still poses a risk of relapse and carcinogenesis after treatment is discontinued. The risk of relapse and carcinogenesis after discontinuation of treatment remains, and complete elimination of the virus has not yet been achieved. In addition to the analysis of the clinical course of chronic hepatitis B patients through long-term observational studies, the Group has been conducting an integrated host-virus gene interaction analysis using cell and mouse infection models and clinical specimens to elucidate the molecular mechanisms involved in persistent HBV infection and carcinogenesis. Based on these results, we aim to overcome HBV infection by establishing therapies that target cccDNA and embedded HBV and that achieve high rates of functional cure or complete elimination of HBV, and by promoting the development of biomarkers that can predict therapeutic efficacy and prognosis.
Identification of biomarkers predictive of carcinogenic risk and novel disease progression in patients with chronic hepatitis B
In a clinical study, a long-term follow-up analysis using a large multicenter cohort revealed that the risk of developing hepatocellular carcinoma (HCC) persists at an annual rate of approximately 1% in patients with chronic hepatitis B under NUC treatment, even in non-cirrhotic cases, more than 10 years after the start of treatment (2). This result indicates that the risk of carcinogenesis is not completely eliminated by NUC treatment, suggesting the importance of long-term carcinogenesis surveillance. Furthermore, we found that GDF15, a novel biomarker candidate, can predict HCC development with high accuracy, indicating that it may be a new risk stratification tool that complements conventional indicators (4). These results are expected to lead to the development of carcinogenesis prediction and surveillance strategies based on personalized medicine.
Immune response and innate immune variation during HBV infection
Under HBV infection, the virus-specific immune response is reduced and the function of immunocompetent cells is altered, which are closely related to the establishment of persistent viral infection. Our group has shown that virus-specific CD8⁺T cells exhibit an exhausted phenotype and increase in frequency in a mouse model of chronic HBV infection (3). Furthermore, we have shown that the phenotype and function of these exhausted T cells are associated with serum HBs antigen levels and HBV DNA, suggesting that immune status is closely related to pathogenesis. We also showed that during co-infection with hepatitis C virus (HCV), the intracellular innate immune response via the RIG-I like helicase pathway induced by HCV infection is activated and HBV replication is suppressed in human hepatocytes (10). These findings support the efficacy of immunomodulatory therapies targeting restoration of T cell function and activation of innate immunity, and are expected to lead to the development of novel therapies aimed at functional cure.
Vulnerability factors for HBV-associated liver cancer
In HBV-associated hepatocellular carcinoma (HCC), specific molecular abnormalities underlying viral infection are thought to be involved in tumor development and progression. The group has shown that in HBV-associated HCC, suppression of INSIG2, a molecule that supports tumor cell survival and proliferation, reduces the proliferative potential of tumor cells and inhibits cell growth. Furthermore, they showed that INSIG2 may contribute to tumor maintenance through cell cycle-related signaling pathways and found that it is a vulnerability factor that could be a potential therapeutic target in HBV-associated HCC (5). Targeting these molecules will enable the development of therapeutic strategies specific to HBV-associated hepatocellular carcinoma based on a mechanism of action different from that of conventional therapy, and is expected to lead to the creation of new treatment options for refractory cases.
Development of new treatments
As part of the development of new therapeutic approaches, the group has been working on CRISPR/Cas9 gene editing therapy targeting cccDNA, which is at the root of persistent HBV infection, and has shown that it is possible to reduce the amount of cccDNA and suppress HBV replication in an infection model using human hepatocytes (7). Furthermore, we showed that the combination of a capsid synthesis inhibitor and interferon (IFN) synergistically enhances the intracellular innate immune response and potently suppresses HBV replication and viral antigen production (8). In addition, FDA compounds that inhibit HBs antigen secretion were identified by library screening (1). Combining these multiple approaches is expected to establish a multifaceted therapeutic strategy aimed not only at inhibition of viral replication but also at functional cure and complete elimination.
Future Prospects: Conquering HBV Infection through Translational Research that Traverses the Basic and Clinical Levels
Ultimately, our goal is to establish novel therapeutic strategies targeting the molecular and immunoregulatory mechanisms underlying persistent HBV infection to achieve complete elimination of HBV and suppression of liver carcinogenesis. Through translational research that goes back and forth between basic and clinical research, we will continue to take on the challenge of overcoming HBV infection.
Major Papers
- Shimoda A, Murai K, Hikita H, Minami S, Miyake T, Kuriki S, Sometani E, Sung J, Shigeno S, Higuchi Y, Maesaka K, Shirai K, Tahata Y, Saito Y, Kodama T, Takahashi T, Suemizu H, Takehara T. Auranofin, identified by FDA-approved drug library screening, inhibits HBs antigen secretion via lysomal damage Takahashi T, Suemizu H, Takehara T. Auranofin, identified by FDA-approved drug library screening, inhibits HBs antigen secretion via lysosomal damage PLoS One. 2026 Jan 16;21(1):e0340023.
- Murai K, Hikita H, Yamada R, Nishimura Y, Miyazaki M, Ishida H, Hosui A, Sakamori R, Tatsumi N, Ohkawa K, Doi Y, Nawa T, Egawa S, Yoshida Y, Nozaki Y, Imanaka K, Nakahara M, Sakakibara M, Yakushijin T, Ogawa H, Usui T, Matsumoto K, Oze T, Kuriki S, Sometani E, Sung J, Shimoda A Tahata Y, Saito Y, Kodama T, Tatsumi T, Yamada T, Takehara T. The Risk of Developing Hepatocellular Carcinoma Persists in Chronic Hepatitis B Patients Even After the Long-Term Administration of Nucleos(t)ide Analogs. Hepatol Res. 2025 Sep;55(9):1228-1238.
- Shigeno S, Kodama T, Murai K, Motooka D, Fukushima A, Nishio A, Hikita H, Tatsumi T, Okamoto T, Kanto T, Takehara T. Intrahepatic Exhausted Antiviral Immunity in an Immunocompetent Mouse Model of Chronic Hepatitis B. Cell Mol Gastroenterol Hepatol. 2025;19(1):101412.
- Sometani E, Hikita H, Murai K, Toyoda H, Tanaka S, Oze T, Sung J, Shimoda A, Fukuoka M, Shigeno S, Fukutomi K, Shirai K, Tahata Y, Saito Y, Nishio A, Furuta K, Kodama T, Sakamori R, Tatsumi T, Mita E, Umezawa A, Tanaka Y, Takehara T. Kodama T, Sakamori R, Tatsumi T, Mita E, Umezawa A, Tanaka Y, Takehara T. High serum growth differentiation factor 15 is a risk factor for the occurrence of Hepatol Res. 2025;55(1):22-33.
- Fukuoka M, Kodama T, Murai K, Hikita H, Sometani E, Sung J, Shimoda A, Shigeno S, Motooka D, Nishio A, Furuta K, Tatsumi T, Yusa K, Takehara T. Genome-wide Cancer Sci. 2024 Mar;115(3): 859-870. 859-870.
- Murai K, Hikita H, Kodama T, Kaibori M, Nishimura Y, Tatsumi T, Yamada T, Kanto T, Mochida S, Takehara T. The impact of the COVID-19 pandemic on The impact of the COVID-19 pandemic on hepatocellular carcinoma diagnosis and treatment in Japan: A multicenter collaborative observational study. Hepatol Res. 2024 May;54(5):439-450.
- Murai K, Kodama T, Hikita H, Shimoda A, Fukuoka M, Fukutomi K, Shigeno S, Shiode Y, Motooka D, Higuchi Y, Miyakawa K, Suemizu H, Ryo A, Tahata Y, Makino Y, Takehara T. Inhibition of nonhomologous end joining-mediated DNA repair enhances anti-HBV CRISPR therapy. Yamada R, Sakamori R, Tatsumi T, Takehara T. Inhibition of nonhomologous end joining-mediated DNA repair enhances anti-HBV CRISPR therapy. Commun. 2022 Sep;6(9):2474-2487.
- Fukutomi K, Hikita H, Murai K, Nakabori T, Shimoda A, Fukuoka M, Yamai T, Higuchi Y, Miyakawa K, Suemizu H, Ryo A, Yamada R, Kodama T, Sakamori R, Tatsumi T, Takehara T. Takehara T. Capsid Allosteric Modulators Enhance the Innate Immune Response in Hepatitis B Virus-Infected Hepatocytes During Interferon Administration. Administration. Hepatol Commun. 2022 Feb;6(2):281-296.
- Yamai T, Hikita H, Fukuoka M, Fukutomi K, Murai K, Nakabori T, Yamada R, Miyakawa K, Watashi K, Ryo A, Kodama T, Sakamori R, Tatsumi T, Takehara T. SIRT1 enhances hepatitis virus B transcription independent of hepatic autophagy. Biochem Biophys Res Commun. 2020 Jun 18;527(1):64-70.
- Murai K, Hikita H, Kai Y, Kondo Y, Fukuoka M, Fukutomi K, Doi A, Yamai T, Nakabori T, Fukuda R, Takahashi T, Miyakawa K, Suemizu H, Ryo A, Yamada R, Kodama T, Sakamori R, Tatsumi T, Takehara T. Hepatitis C virus infection suppresses hepatitis B virus replication via the RIG-I-like helicase pathway. Sakamori R, Tatsumi T, Takehara T. Hepatitis C virus infection suppresses hepatitis B virus replication via the RIG-I-like helicase pathway. 2020 Jan 22;10(1):941.
- Nakabori T, Hikita H, Murai K, Nozaki Y, Kai Y, Makino Y, Saito Y, Tanaka S, Wada H, Eguchi H, Takahashi T, Suemizu H, Sakamori R, Hiramatsu N, Tatsumi T, Takahara T. Takehara T. Sodium taurocholate cotransporting polypeptide inhibition efficiently blocks hepatitis B virus spread in mice with a humanized liver. Rep. 2016 Jun 9;6:27782.
