Supercritical Fluid
A supercritical fluid (SCF) is a fluid in which both temperature and pressure exceed the critical point of the gas-liquid phase transition; it can be considered an ideal solvent that combines the solubility of a liquid with the diffusivity of a gas. Because the properties of the solvent can be freely adjusted by controlling temperature and pressure, it is regarded as a highly functional medium capable of delivering solvent performance tailored to specific applications. Furthermore, technologies utilizing supercritical fluids are advantageous in terms of environmental impact and cost, as they can reduce the amount of organic solvents used. Supercritical fluids have been used in extraction, separation and purification, and reactions; in addition, applications that leverage the characteristics of supercritical fluids are being explored in a wide range of fields, including cleaning, dyeing, pulverization, and foaming.
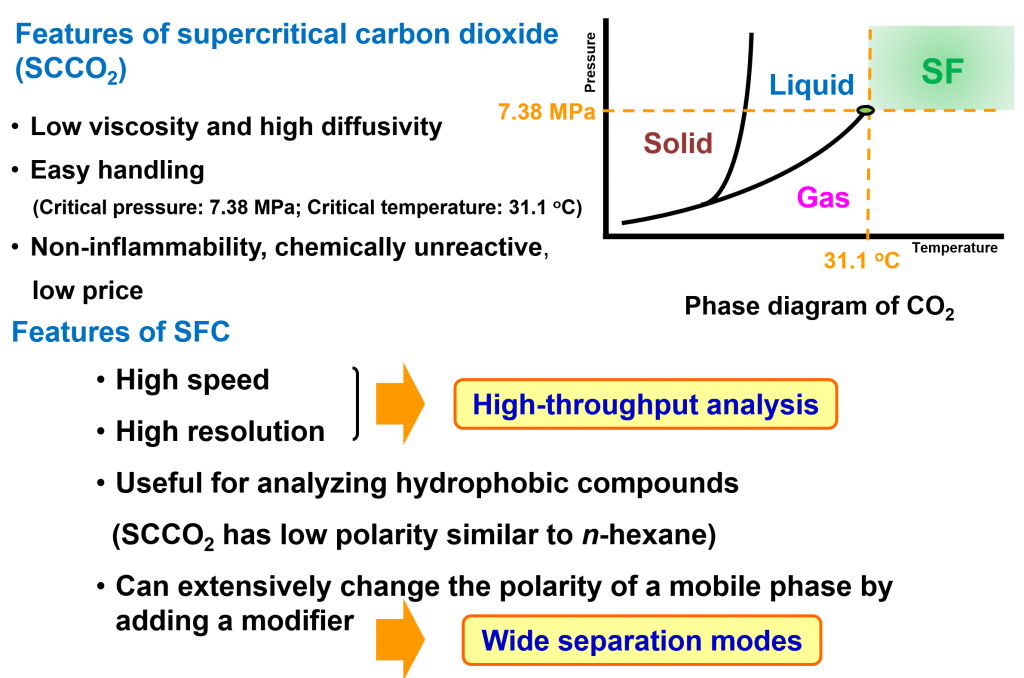
Among supercritical fluids, supercritical carbon dioxide (SC-CO₂) is the most commonly used due to its properties: (1) relatively low critical temperature (31.1 °C) and critical pressure (7.38 MPa), (2) ability to dissolve many organic compounds, (3) chemical inertness, (4) it is non-toxic and inexpensive. The Baba Laboratory has been exploring the use of SC‒CO₂ in the “extraction” and “separation” processes of metabolome analysis¹). Extraction operations using supercritical fluids as the extraction medium are called supercritical fluid extraction (SFE), and chromatography using supercritical fluids as the mobile phase is called supercritical fluid chromatography (SFC). Furthermore, supercritical fluid extraction–supercritical fluid chromatography (SFE–SFC), which connects SFE and SFC online, is a technique that enables extraction and analysis to be performed in a single step.
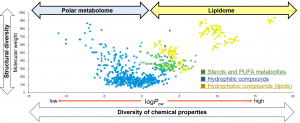
Since the polarity of SC-CO₂ is considered to be comparable to that of hexane, it is regarded as useful for the separation and analysis of hydrophobic compounds. Initially, the mainstream approach involved using 100% carbon dioxide as the mobile phase and applying gradients by varying temperature and pressure; however, in recent years, it has become more common to use subcritical fluids created by adding organic solvents such as methanol to SC-CO₂ as modifiers. This is because adding a modifier increases the solubility of polar compounds in the mobile phase, enabling the simultaneous separation and analysis of compounds with a wide range of physical properties.
As a practical application of supercritical fluid technology, we have been developing a technique for the simultaneous analysis of lipid molecules (lipome analysis) using SFC/MS. By connecting a quadrupole orbitrap mass spectrometer—capable of high-resolution mass measurements and MS² analysis—to the SFC system, we have successfully identified and quantified more than 500 lipid species from mouse plasma extracts²). Furthermore, SFE is a method in which the supercritical fluid (SCF) is added to a sample containing the target component, and extraction is performed by utilizing differences in solubility. Compared to conventional organic solvent extraction methods, SFE offers advantages such as high extraction efficiency and the ability to perform extraction under mild conditions in the dark and in an oxygen-free environment, resulting in a low environmental impact. Therefore, we have also investigated an online SFE-SFC/MS metabolic profiling method that maximizes the characteristics of SFE and the advantages of SFC/MS. Online SFE-SFC/MS is a technology applicable to filter paper blood samples for newborn mass screening, diagnosis of hereditary diseases, and biomarker screening. When phospholipid profiling using online SFE-SFC/MS was performed on filter paper blood prepared from a small amount of mouse plasma (3 μL), we successfully quantified 134 types of phospholipids³).
Supercritical fluid technology is a novel technique that expands the range of solvents available for the “extraction” and “separation” of compounds. While its usefulness is gradually gaining recognition, we intend to continue developing new analytical methods and applications that leverage the properties of supercritical fluids. In our article on analytical technology development, we introduce a new analytical method for quantitative lipidomics, so we encourage you to read it.
References
- Bamba T, Lee JW, Matsubara A et al : Metabolic profiling of lipids by supercritical fluid chromatography/mass spectrometry. J Chromatogr A 1250, 212–219 (2012).
- Yamada T, Uchikata T, Sakamoto S et al : Supercritical fluid chromatography/orbitrap mass spectrometry based lipidomics platform coupled with automated lipid identification software for accurate lipid profiling. J. Chromatogr. A 1301, 237–242 (2013).
- Uchikata T, Matsubara A, Fukusaki E et al : High-throughput phospholipid profiling system based on supercritical fluid extraction-supercritical fluid chromatography/mass spectrometry for dried plasma spot analysis. J. Chromatogr. A 1250, 69–75 (2012).