C) Molecular mechanisms of the function of BST-1/CD157 and lymphopoiesis
-------Elucidation of roles and mechanisms of novel GPI-anchored
molecule, BST-1(CD157) and Identification and cloning of BST-1
ligand.------- Key words: ADP ribosyl cyclase, ectoenzyme, CD38,
lymphocyte development, macrophage function, antibody production,
germinal center, stromal cells, rheumatoid arthritis. Methods: FACS
analysis, cell sorting, gene targetting, transgenic mice, monoclonal
antibody, expression cloning, histochemistry, etc
(publication)
Katsuhiko Ishihara, Yoshiki Okuyama, Byung OK Lee, Motoyuki Itoh, Kayoko Nishikawa, and Toshio Hirano. Leucocyte typing VI, 1086-1089, 1997
We previously showed that bone marrow (BM) stromal cell lines derived from patients with rheumatoid arthritis (RA) have a high supporting ability of the growth of a pre-B cell line[1]. We made a monoclonal antibody (mAb),RF3, which recognizes a surface molecule expressed on a RA-derived BM stromal cell line which can support a pre-B cell line growth[2]. Expression cloning revealed that RF3 recognizes a novel molecule, which we designate BST-1 (BM stromal cell antigen 1), that facilitates the growth of a pre-B cell line, DW34. BST-1 is a glycosyl phosphatidylinositol (GPI)-anchored ectoenzyme having homology with Aplysia ADP-ribosyl cyclase and CD38[2]. BEC7 is another anti-BST-1 mAb established by immunizing mice with recombinant soluble BST-1 protein[3]. In the 6th HLDA workshop, to search mAbs which react with BST-1 we examined 329 mAbs submitted to non-lineage, endothelial and myeloid cell sections utilizing a CHO cell transfectant expressing human BST-1(CHO-BST-1). We found that a mAb M51(anti-Mo5) reacted with CHO-BST-1 but not with control CHO cells(Table 1, Fig.1A, B). Mo5 is a myeloid antigen originaly reported by Todd et al. [4] who made anti-Mo5 mAb by immunizing a mouse with leukemic cells from a patient with acute monocytic leukemia. Thus three mAbs, E060/N-L118(RF3), E061/N-L117(BEC7), and M51(Mo5) are clustered in a new CD.
Cellular Expression
Analysis with RF3 or BEC7 revealed that BST-1 is expressed on BM stromal cell lines, synovial cell lines, HUVEC, follicular dendritic cell lines, myelomonocytic cell lines, peripheral granulocytes, monocytes and in vitro differentiated macrophages[2, 3, 5]. Todd et al. showed that Mo5 is expressed on human peripheral blood monocytes, neutrophils, and all myeloperoxidase-positive BM myeloid precursors but not nonmyeloid cells in peripheral blood and BM[4]. Mo5 is expressed by the 74% of patients with acute monocytic leukemia (FAB group M4, M5) and 50% of patients with acute granulocytic leukemia (FAB group M1, M2, and M3)[4]. Expression of Mo5 on neutrophils is upregulated by the treatment with calcium ionophore, whereas that on neutrophils and monocytes is downmodulated by the treatement with divalent anti-Mo5 mAb[4]. In the blind panel analysis of the 5th HLDA workshop, it was reported that Mo5 is expressed on eosinophils and basophils from peripheral blood and on a mast cell line, HMC-1[6]. Furthermore, fMLP upregulates Mo5 expression on neutrophils and basophils but not monocytes. PAF upregulates Mo5 expression on eosinophils, and TNFa upregulates, while GM-CSF downregulates its expression on U937.
Immunochemistry
BST-1 expressed on BM stromal and joint synovial cell lines is a single-chain 42-45kDa glycoprotein anchored into the membrane via GPI [7]. Deglycosylated BST-1 is 31kDa[8]. Analysis utilizing anti-Mo5 mAb showed that Mo5 is synthesized as a 41-43kDa precursor which is converted to the mature 50kDa glycoprotein within 1-4 hours in both U937 and PMN[9].
Molecular cloning
The human BST-1 cDNA encodes a protein consisting of 290 aminoacids attached to the GPI-anchor. The deduced amino acid sequence of BST-1 has 33% homology with human CD38 and 26% homology with Aplysia ADP-ribosyl cyclase[2]. The positions of nine cysteine residues of BST-1 are completely conserved among CD38 and the cyclase. Molecular cloning of murine BST-1 and BP-3, a marker for early B, myeloid and reticular cells, revealed that both are identical molecules[10-13]. Furthermore, the cloning of murine pre-T cell antigen recognized by mAb IF-7 showed that it is identical with BST-1/BP-3[14]. Murine and rat BST-1 shows 71% and 72% homology of amino acid sequence with human BST-1, respectively[10,15].
Chromosome location
We previously mapped the human Bst-1 gene to 14q32.3[2] but we have recently re-assigned it to 4p15 (Inazawa et al. manuscript in preparation). The gene located in 14q32.3 is most likely the pseudogene.
Epitope analysis
To analyze the epitope recognized by E060/N-L118(RF3), E061/N-L117(BEC7) and M51(anti-Mo5), we treated CHO-BST-1 cells with M51 ascites, then stained the cells with either biotinylated RF3 or biotinylated BEC7 together with FITC-streptavidin(Fig.1C,D). The pretreatment of the cells with M51 blocked the binding of RF3 and BEC7, suggesting that these three mAbs recognize very close epitopes on BST-1.
Function
Supporting the growth of pre-B cells : Stable transfectants of BALB3T3 expressing human BST-1 facilitate the growth of a murine pre-B cell line DW34[2].
Function of enzymatic activities: BST-1 is bifunctional ectoenzyme having both ADP-ribosyl cyclase and cyclic ADP-ribose hydrolase activities[8]. ADP-ribosyl cyclase catalyzes the synthesis of cyclic ADP-ribose from NAD, then cyclic ADP-ribose is hydrolized to ADP-ribose. Cyclic ADP-ribose is known to serve as an endogenous second messenger that regulates the release of Ca2+ ion from endoplasmic reticulum[16], but the roles of extracellular cyclic ADP-ribose and ADP-ribose are unkown.
Function as a receptor: Closslinking of BST-1 with polyclonal anti-BST-1 antibody induces tyrosine phospohrylation of 130kDa protein in U937 and THP-1[3]. Crosslinking of BST-1 expressed on CHO-BST-1 transfectant induces tyrosine phospohrylation of 130kDa protein, dephosphorylation of 100kDa protein, and growth inhibition[3]. Thus BST-1 could function as a receptor being capable of generating signal transduction.
Pathophysiolosical roles in RA: Enhanced expression of BST-1 on RA-derived BM stromal cell lines is observed[2]. Furthermore elevated levels of serum soluble BST-1 at 30-50 times higher concentration is observed in 7% of RA cases, all of which belong to severe RA[7]. Since BST-1 functions as a receptor, overexpression of membrane or soluble BST-1 may have the pathophysiological roles by modifying the cellular function in the joints and BM, the major lesions of RA.
Function as a myeloid antigen: Analysis using anti-Mo5 mAb in the myeloid blind panel of the 5th HLDA, revealed several functional roles in myeloid cells[6]. Mo5 inhibits transepitherial migration of neutrophils in the apical-to-basolateral direction but not opposit direction. Anti-Mo5 mediates the superoxide (O2-)-production in U937. Anti-Mo5 suppresses PMN phagocytosis of S.aureus.
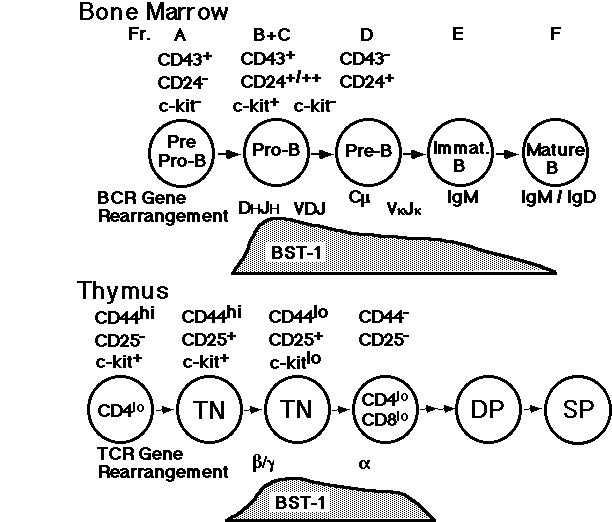
Function in murine lymphoid progenitors: Anti-pre-T cell mAb IF-7, recognizing BST-1/BP-3 antigen, has a synergistic effect on anti-CD3 induced growth, and facilitates the development of abTCR+ T cells in fetal thymic organ culture[14]. Analysis with anti-murine BST-1 mAb G12 showed that the begining of BST-1 expression on B and T cell progeniters coincides with the stage when the gene rearrangement of immunoglobulin m chain and T cell receptor b chain, respectively [17].These results suggest that murine BST-1 plays roles around the critical stage in early lymphoid development.
Reference
1) Kaisho,T., Oritani, K., Ishikawa, J., Tanabe, M., Muraoka, O., Ochi, T., et al. Jounal of Immunology 149, 4088-95 (1992).
2) Kaisho,T., Ishikawa, J., Oritani, K., Inazawa, J.,Tomizawa, H., Muraoka, O., et al. Proceedings of National Academic Sciety of USA 91, 5325-9 (1994).
3) Okuyama, Y., Ishihara, K., Kimura, N., Hirata, Y., Sato, K., Itoh, M., et al. Biochemical and Biophysical Research Communications in press.
4) Todd, R.F.III., Roach, J.A., Arnaout, M.A. Blood 65, 964-73 (1985).
5) Clark, E.C., Grabstein, K.H., Gown, A.M., Skelly, M., Kaisho, T., Hirano, T. et al. Journal of Immunology 155, 545-55 (1995).
6) In Leucocyte typing V (ed. R. F. Todd III), pp991-1093. Oxford University Press, Oxford (1995).
7) Lee, B.O., Ishihara, K., Denno, K., Kobune, Y., Itoh, M., Muraoka, O., et al. Arthritis & Rheumatism 39, 629-37 (1996).
8) Hirata, Y., Kimura, N., Sato, K., Ohsugi, Y., Takasawa, S., Okamoto, H. et al. FEBS Letters 356, 244-8 (1994).
9) Goldstein, S.C., and ToddR.F.III. Tissue Antigens 41, 214-8 (1993)
10) Itoh, M., Ishihara, K., Tomizawa, H., Tanaka, H., Kobune, Y., Ishikawa, J. et al. Biochemical and Biophysical Research Communications 203, 1309-17 (1994).
11) Dong, C., Wang, J., Neame, P. and Cooper, M. D. International Immunology 6, 1353-60 (1994).
12) McNagny, K.M., Cazenave, P-A., and Cooper, M. D. Journal of Immunology 141, 2551-6 (1988).
13) McNagny, K.M., Bucy, R.P., and Cooper, M. D. European Journal of. Immunology 21, 509-15 (1991).
14) Vicari, A. P., Bean, A. G. D. and Zlotnik, A. International Immunology 8, 183-91 (1996).
15) Furuya, Y., Takasawa, S., Yonekura, H., Tanaka, T., Takahara, J., and Okamoto, H. Gene 165, 329-30 (1995).
16) Lee, H.C., Galione, A., and Walseth, T. F. Vitamins and hormones 48, 199-257 (1994).
17) Ishihara, K., Kobune, Y., Okuyama, Y., Itoh, M., Lee, B.O., Muraoka, O. et al. International Immunology 8, 1395-404 (1996).