|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
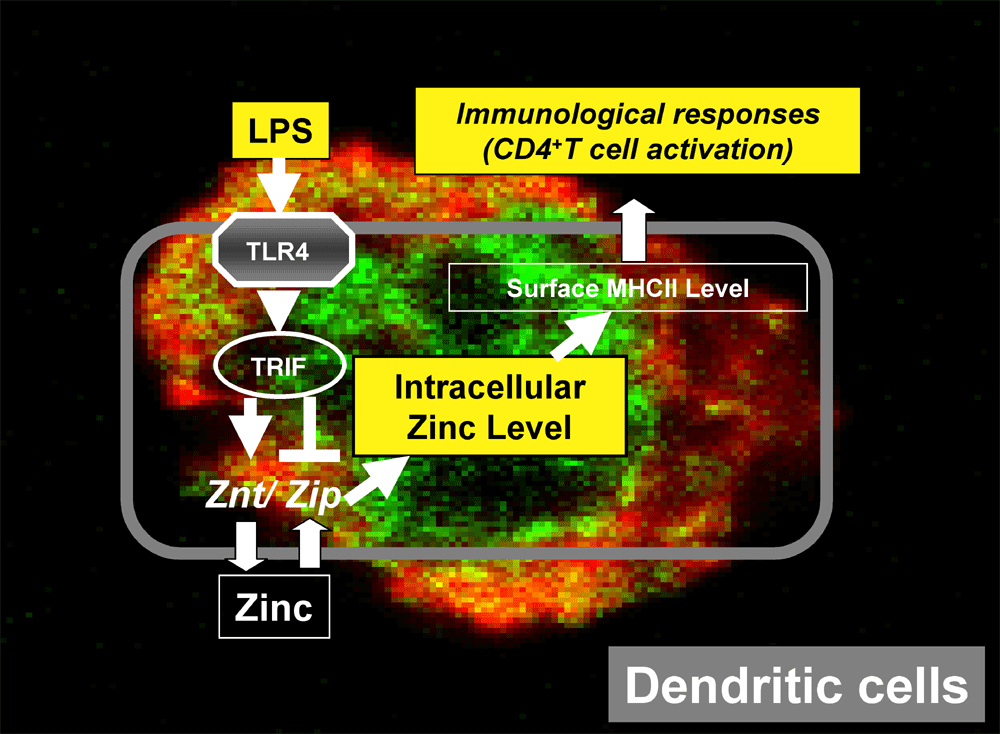
|
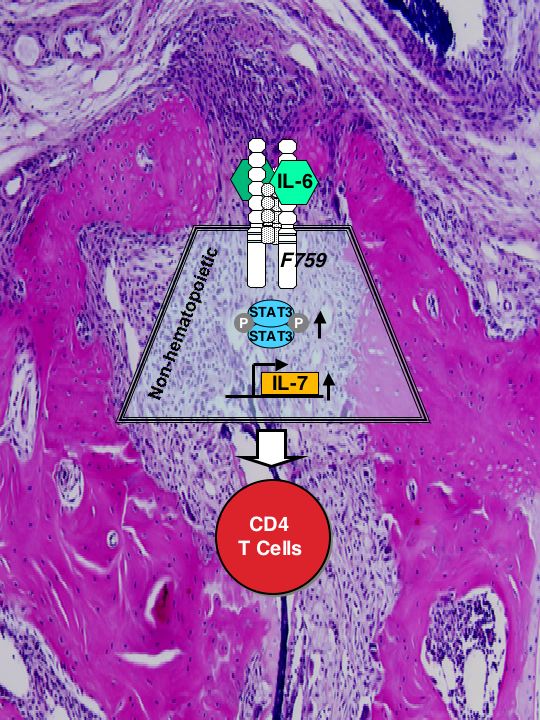
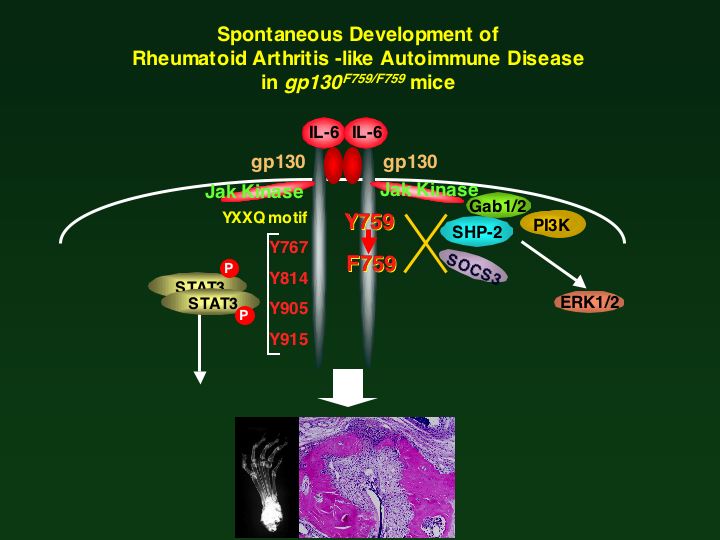
Hyperresponsiveness to the interleukin (IL)-6 family of cytokines triggers a spontaneous rheumatoid arthritis (RA)-like disease in mice (Atsumi et al, JEM 2002)(PubMed). Now, Sawa and colleagues show that excessive IL-6 signaling drives hyperproliferation of CD4+ T cells, which then attack the joints. IL-6 has been implicated in RA and other T cell-driven autoimmune diseases. Indeed, a previous study by this group showed that an activating mutation in the gp130 subunit of the IL-6 receptor caused a lymphocyte-driven arthritis in mice. But the mechanism was unclear. The authors now show that disease development in these mice depends on CD4+ T cells, but not on cytolytic CD8+ T cells or antibody-producing B cells. The CD4+ cells did not appear to cause disease because of an affinity for joint-specific antigens. Rather, the cells simply proliferated excessively in the mutant mice. This hyperproliferation was not the fault of the T cell, as wild-type CD4+ T cells also multiplied excessively and caused disease when transferred into irradiated mutant mice. Rather, the gp130 mutation caused nonhematopoietic cells to produce excess IL-7--a growth factor that triggers T cell proliferation. This is the first evidence that IL-6 family cytokines can trigger IL-7 production. These data suggest that IL-6, which is elevated in the serum and joints of patients with RA, might exacerbate disease by inducing IL-7 and thus driving T cell activation. What causes the overstimulated T cells to attack the joints in the first place remains a mystery. J Exp. Med. In This Issue Autoimmunity: Cytokine partnership to destruction: Researchers reporting in The Journal of Experimental Medicine have identified the mechanism behind a form of spontaneous autoimmune arthritis that occurs in mice with a mutation in the interleukin-6 (IL-6) receptor subunit gp130. Intriguingly, it turns out that signalling by this mutant IL-6 receptor in non-haematopoietic cells ----, taken from Nature Reviews Immunology 6:493, 2006, July Issue, Research Highlights.
|
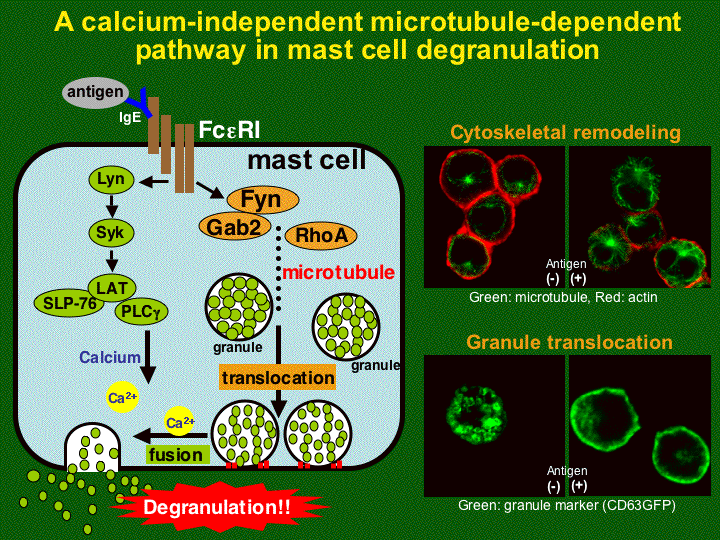
Nishida, K., S. Yamasaki, Y. Ito, K. Kabu, K. Hattori, T. Tezuka, H. Nishizumi, D. Kitamura, R. Goitsuka, R.S. Geha, T. Yamamoto, T. Yagi, T. Hirano. FceRI-mediated mast cell degranulation requires calcium-independent microtubule-dependent translocation of granules to the plasma membrane. J. Cell Biol. 170:115-126, 2005 (PubMed). (JCB) (In This Issue)
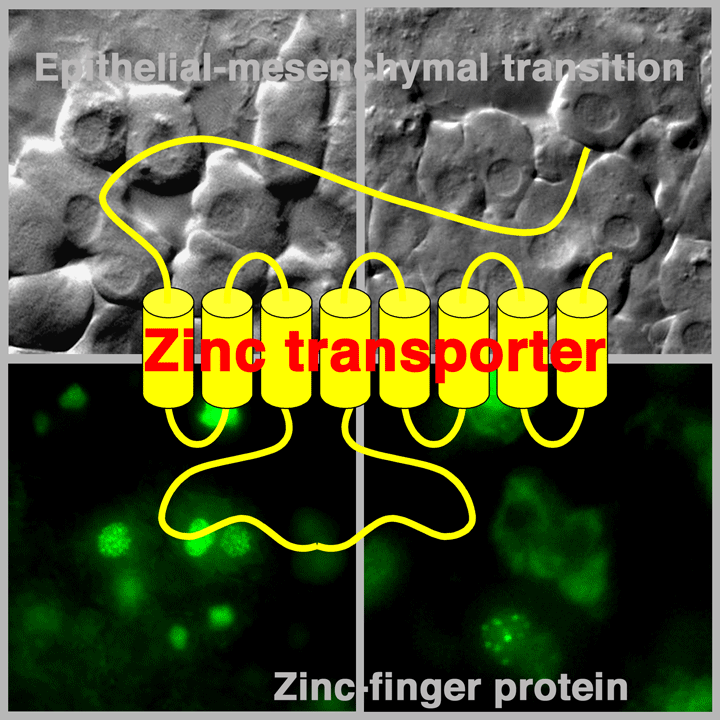
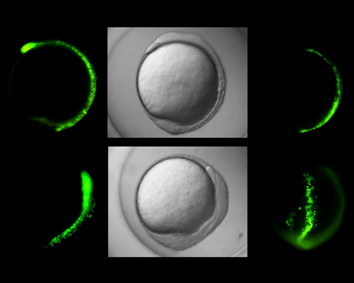
ゼブラフィシュ受精卵の初期発生における細胞運動制御機構を解析することにより、細胞の可動性獲得の鍵となる亜鉛要求性転写因子の活性を制御する因子を同定することに成功しました。この成果は、これまで謎とされてきた亜鉛輸送体による転写因子の活性制御機構を解明した世界で初となる研究成果です。今後細胞内亜鉛輸送機構を詳細に解析することによって、人間の体の形作りのメカニズムに迫るとともに、癌転移予防薬の確立につながるものと期待されます。
Atsumi, T*., K. Ishihara*, D. Kamimura, H. Ikushima, T. Ohtani, S. Hirota, H. Kobayashi, S-.J. Park, Y. Saeki, Y. Kitamura, and T. Hirano. (*equal contribution). A point mutation of Tyr-759 in interleukin 6 family cytokine receptor subunit gp130 causes autoimmune arthritis. J. Exp Med. 196: 979-990, 2002 (PubMed)
本論文はNature Immunology 11月号で紹介された(Nature Immunology Nov. issue, 2002)
Nature Immunology 3, 1060 (2002) Immunology highlights from the recent literature
Pointed autoimmunity: The IL-6 receptor consists of alpha chain paired with the subunit gp130. By introducing a single point mutation of gp130 (Y759--->F759), Hirano and colleagues generated SHP-2 signal-deficient mice. They report in the Journal of Experimental Medicine that these mice spontaneously develop rheumatoid arthritis. Mutant mice produce autoantibodies and have greater numbers of activated T cells and myeloid cells. Both thymic selection and clonal deletion of peripheral T cells were impaired. Mature T cells exhibited hyperresponsiveness even before disease onset, and IL-6-induced STAT3 activation was prolonged, which enhanced the inhibitory effect of IL-6 on FasL expression. Thus, a point mutation in a cytokine receptor can effect B and T cell abnormalities that cause autoimmunity. These mice may provide a useful animal model for spontaneous rheumatoid arthritis.(Nature Immunology Nov. issue, 2002)
インターロイキン6サイトカイン受容体の変異により自己免疫疾患が自然発症するという画期的な発見であり、自己免疫疾患の分子メカニズムを解明するために非常に有用なモデルです
Yamashita, S., C. Miyagi, A. Carmany-Rampey, T. Shimizu, R. Fujii, A. F. Schier and T. Hirano. Stat3 controls cell movements during zebrafish gastrulation. Dev. Cell. 2: 363-375, 2002.(PubMed) (Evaluations and comments from leading biologists)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
日本分子生物学会 | ||
|
日本癌学会 |
|
Tel 06-6879-3881 |
Fax 06-6879-3889 |